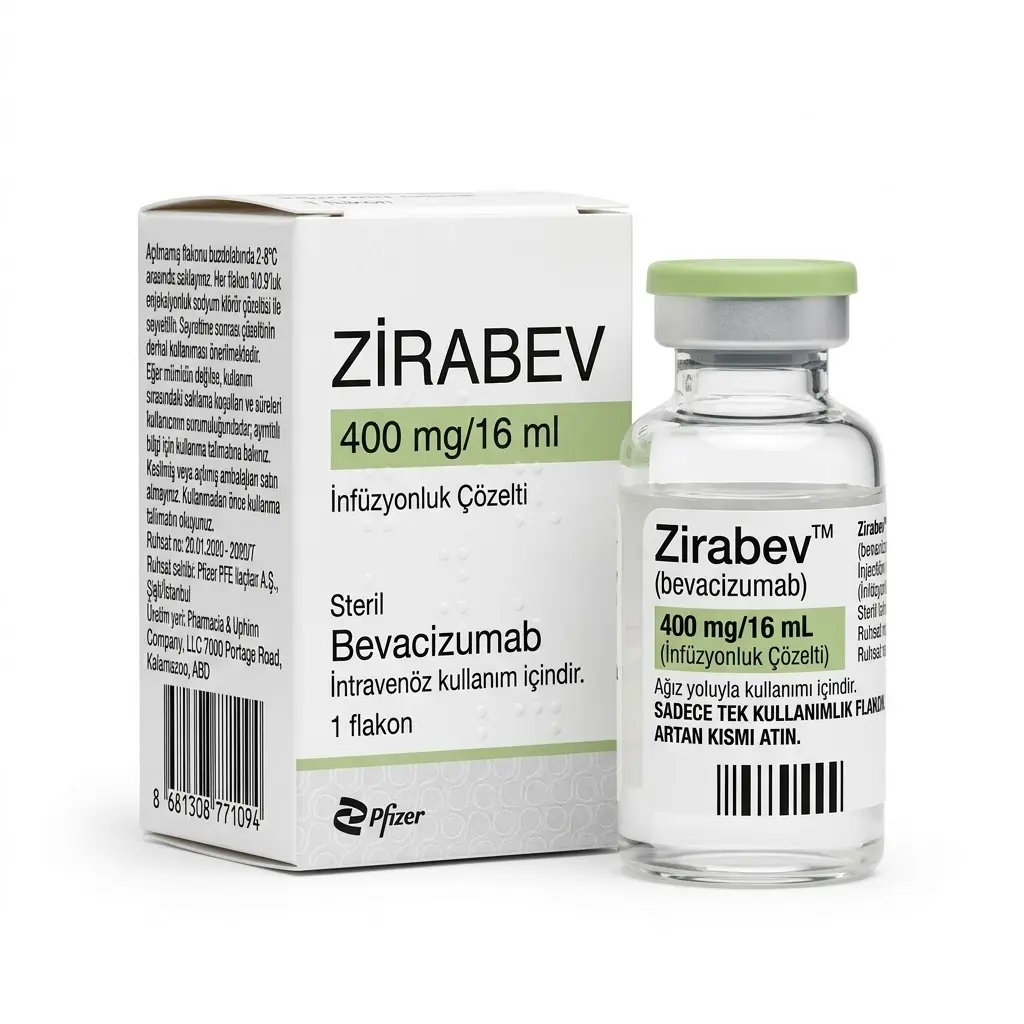
Zirabev 400 Vial — Bevacizumab 400 mg/16 ml (1 Vial) Pfizer
Zirabev 400 mg/16 ml (Bevacizumab) is a highly effective targeted cancer therapy. It is a recombinant humanized monoclonal antibody that binds to the vascular endothelial growth factor (VEGF). By blocking VEGF, Zirabev prevents the formation of new blood vessels that tumors need to grow. Deprived of oxygen and essential nutrients, the tumor is starved and its progression is halted.
Manufacturer: Pfizer (USA). 🇹🇷 Note: This specific product holds official Turkish registration. It is a premium biosimilar to the well-known brand Avastin, manufactured by the global pharmaceutical giant Pfizer for the domestic market of Turkey, ensuring strict adherence to international quality standards.
Key Features:
- ✅ Anti-Angiogenic Effect: Directly targets and cuts off the tumor's blood supply.
- ✅ Broad Applicability: Proven effective in treating a wide variety of solid tumors.
- ❄️ COLD CHAIN REQUIRED: This medication must be stored in a refrigerator at 2°C to 8°C (36°F to 46°F). Do not freeze!
Notice. The information on this page is for reference only and does not replace medical consultation. Always consult a healthcare professional and read the manufacturer's instructions before using any medicine. Self-medication may be dangerous. Information updated: 09.04.2026
Similar products
What Customers Say
No reviews yet
Your review can be the first!