Erlotinib – Targeted Therapy
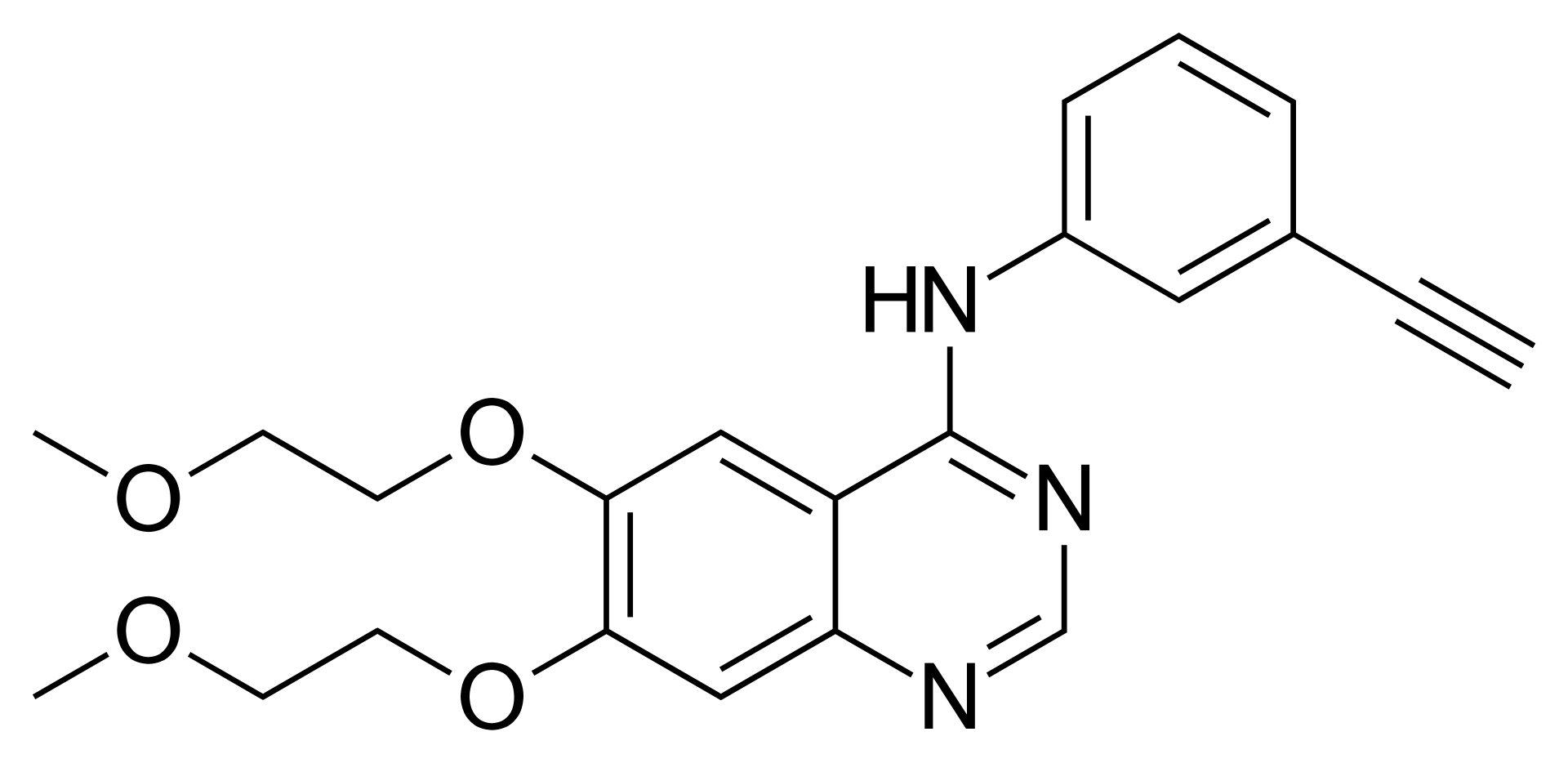
Erlotinib is a potent antineoplastic agent belonging to the class of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors. The drug is a highly selective low-molecular-weight inhibitor designed for targeted therapy of malignant neoplasms whose development is associated with the activation of the HER1/EGFR receptor signaling pathways.
The mechanism of action of erlotinib is based on competitive inhibition of adenosine triphosphate (ATP) binding to the intracellular tyrosine kinase domain of the EGFR receptor. This blocks receptor autophosphorylation and interrupts downstream signaling cascades responsible for cell proliferation, angiogenesis, and tumor survival. The drug is particularly effective in the presence of specific activating mutations in the EGFR gene (exon 19 deletions or L858R substitution in exon 21).
Erlotinib exhibits high bioavailability when administered orally. The systemic effect of the drug allows for efficient suppression of tumor cell growth and the induction of apoptosis, leading to disease stabilization and increased progression-free survival in patients with metastatic forms of cancer.
Indications
Erlotinib is used for the treatment of solid tumors with confirmed molecular genetic status:
- Non-Small Cell Lung Cancer (NSCLC): as first-line therapy and maintenance therapy in patients with locally advanced or metastatic disease harboring activating EGFR mutations.
- Pancreatic Cancer: in combination with gemcitabine for the first-line treatment of patients with metastatic pancreatic adenocarcinoma.
- Advanced NSCLC: as second- or third-line therapy after the failure of at least one prior standard chemotherapy regimen.
- The drug may be prescribed as maintenance treatment for patients with stable disease following four cycles of standard first-line platinum-based chemotherapy.
Dosage and administration
Erlotinib therapy requires constant supervision by an oncologist and adherence to a strict dosing regimen in tablet form.
- In Lung Cancer (NSCLC): the standard dose is 150 mg once daily.
- In Pancreatic Cancer: the recommended dose is 100 mg once daily in combination with intravenous gemcitabine.
- Administration Rules: the drug must be taken strictly on an empty stomach — at least 1 hour before or 2 hours after food intake. Food significantly increases bioavailability, which may lead to unpredictable toxicity.
- Lifestyle Factors: smoking significantly reduces plasma concentrations of erlotinib (by 50–60%) through the induction of metabolic enzymes; therefore, patients are strongly advised to stop smoking.
- Dose Adjustment: if severe side effects or pronounced skin rash develop, the dose is reduced stepwise (by 50 mg) or treatment is temporarily interrupted.