Raltegravir: HIV treatment drugs list & full guide
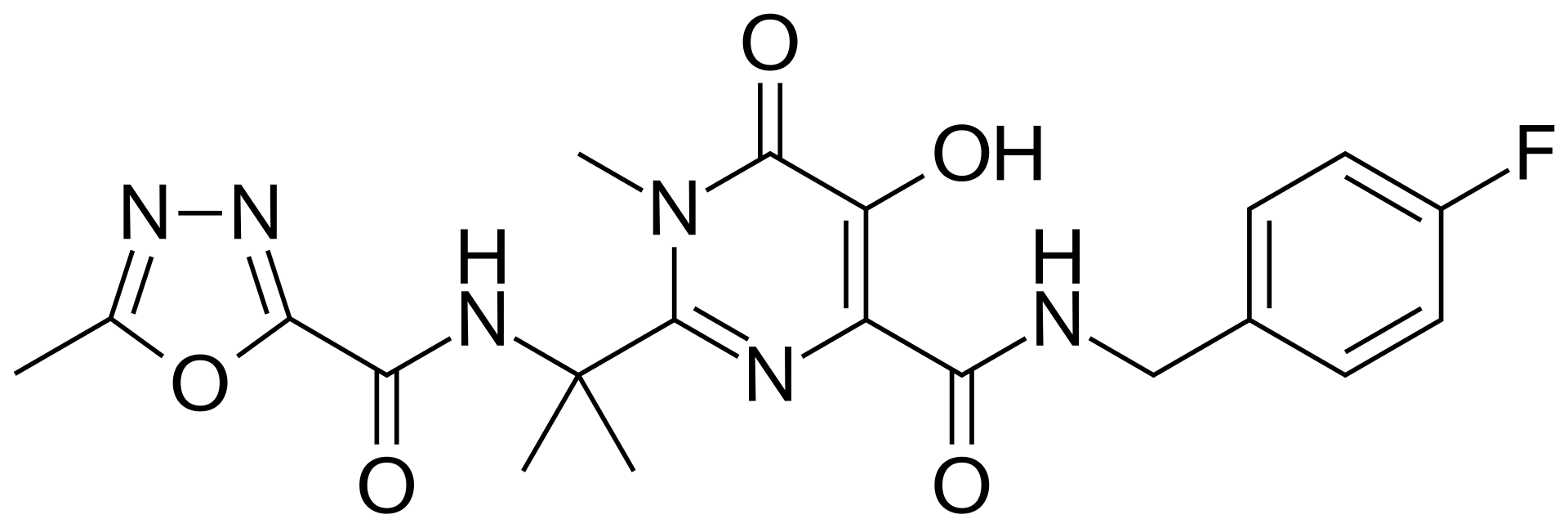
Raltegravir is a pioneering antiretroviral medication and the first approved HIV integrase strand transfer inhibitor (INSTI). It works by blocking the enzyme that allows the virus to integrate its DNA into the human genetic code, effectively preventing viral replication.
It is highly valued for its potency, safety profile, and minimal drug-drug interactions. It is frequently the drug of choice for Post-Exposure Prophylaxis (PEP). On Unifarm, you can find a comprehensive list of available Raltegravir formulations.
Indications
- Treatment of HIV-1 infection in adults, adolescents, and children (as part of combination antiretroviral therapy).
- Used in both treatment-naïve patients and those with experienced resistance to other drug classes.
- Post-Exposure Prophylaxis (PEP): To prevent HIV infection after potential exposure (in combination with other antiretrovirals).
Dosage and administration
Taken orally, with or without food. Tablets should not be chewed or crushed.
Standard Adult Regimens:
- 400 mg twice daily (every 12 hours).
- 1200 mg (two 600 mg tablets) once daily — only for clinically stable patients using the specific high-dose formulation.
Important: Do not take antacids (containing aluminum or magnesium) simultaneously with Raltegravir, as they significantly reduce absorption.