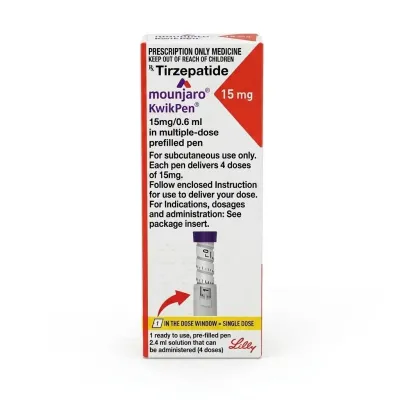
Mounjaro KwikPen 2.5 — Tirzepatide 2.5 mg (1 Pen / 4 Doses)
Mounjaro KwikPen 2.5 mg (Tirzepatide) is an innovative, first-in-class dual-action medication designed for blood sugar control and weight management. Tirzepatide acts as a receptor agonist for two hormones: glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1). This combination allows for more effective reduction of glycated hemoglobin levels, slows gastric emptying, and significantly reduces appetite.
Manufacturer: Eli Lilly. Mounjaro in the KwikPen format is a multi-dose pre-filled pen that ensures convenience and precision of administration. It is an original drug that has undergone extensive clinical trials (the SURPASS program), confirming its superiority over many existing therapies for type 2 diabetes and obesity.
Key Features:
- ✅ Dual Mechanism: Action on both GIP and GLP-1 receptors provides a synergistic effect in weight loss and metabolic normalization.
- ✅ KwikPen Format: One pen contains 4 doses of the drug, which is designed for a full one-month course of initial therapy.
- ✅ Cardioprotective Effect: Positively affects blood pressure and lipid profiles, reducing the risk of cardiovascular complications.
Notice. The information on this page is for reference only and does not replace medical consultation. Always consult a healthcare professional and read the manufacturer's instructions before using any medicine. Self-medication may be dangerous. Information updated: 19.04.2026
What Customers Say
No reviews yet
Your review can be the first!