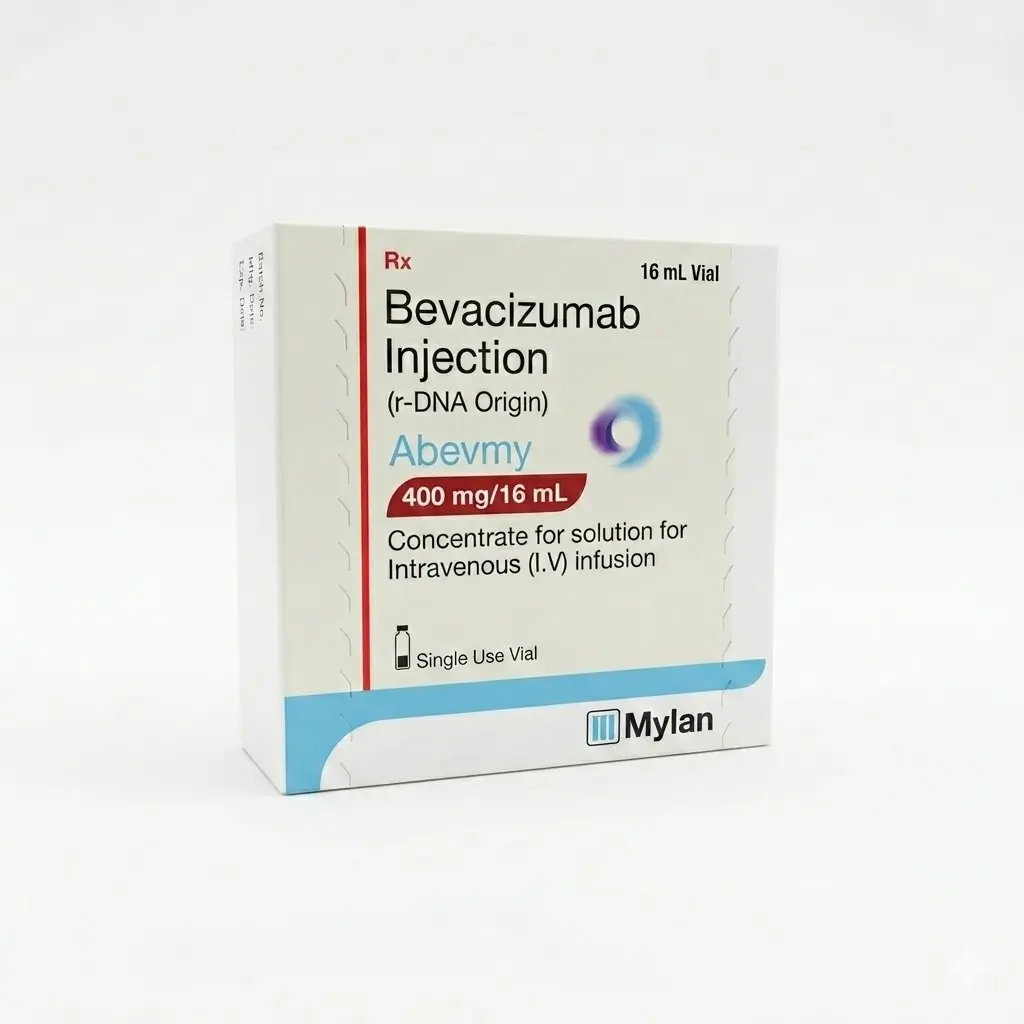
Abevmy 400 Vial — Bevacizumab 400 mg/16 ml (1 Vial) Mylan
Abevmy 400 mg/16 ml (Bevacizumab) is an advanced targeted anti-cancer therapy. It is a monoclonal antibody that selectively binds to the vascular endothelial growth factor (VEGF). By blocking this factor, Abevmy inhibits the development of new blood vessels in the tumor, effectively cutting off its supply of oxygen and nutrients, which stops tumor growth and metastatic spread.
Manufacturer: Mylan (in collaboration with Biocon). 🌍 This drug is a high-quality biosimilar of the original drug Avastin, manufactured under strict international standards for quality and clinical safety. It has undergone rigorous testing to prove therapeutic equivalence to the reference product.
Key Features:
- ✅ Targeted Mechanism: Specifically attacks the tumor's blood supply network while sparing healthy organs.
- ✅ Broad Spectrum: Proven effective for various types of solid tumors.
- ❄️ STRICT TEMPERATURE CONTROL: As a biological product, Abevmy requires continuous "cold chain" management. It must be stored in a refrigerator at 2°C to 8°C. Do not freeze or expose to heat.
Notice. The information on this page is for reference only and does not replace medical consultation. Always consult a healthcare professional and read the manufacturer's instructions before using any medicine. Self-medication may be dangerous. Information updated: 10.04.2026
Similar products
What Customers Say
No reviews yet
Your review can be the first!