Entecavir – Chronic Hepatitis B Therapy
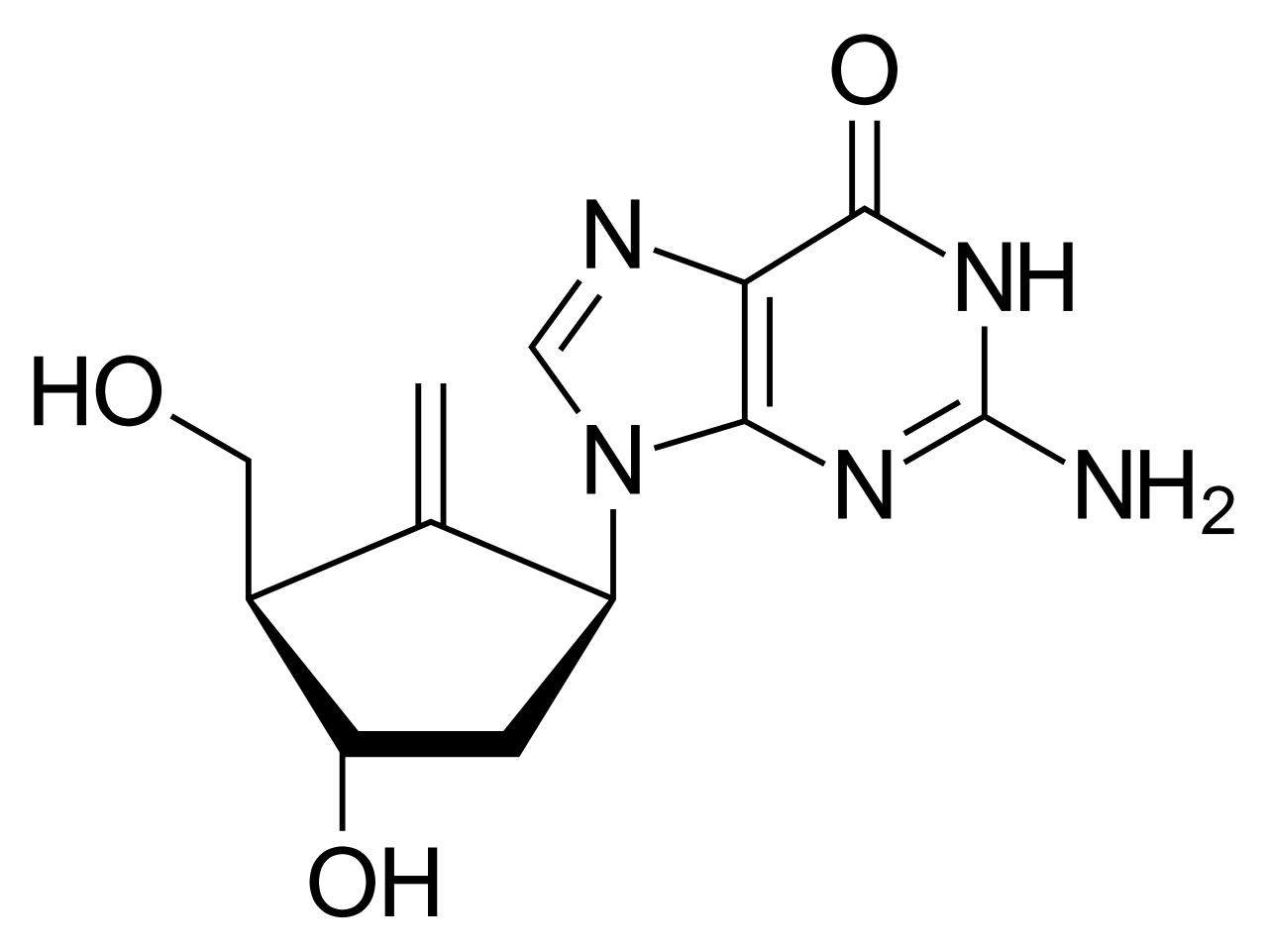
Entecavir is a potent antiviral medication belonging to the nucleoside (guanine) analog class. It is specifically designed to inhibit the replication of the hepatitis B virus (HBV) and is recognized as a first-line therapy choice due to its high potency and low genetic barrier to resistance.
The mechanism of action involves the inhibition of HBV DNA polymerase. Entecavir effectively blocks three functional stages of the viral life cycle: HBV polymerase priming, reverse transcription of the negative strand from pregenomic mRNA, and synthesis of the positive strand of HBV DNA. Inside the cell, entecavir is phosphorylated to its active triphosphate form, which competes with the natural substrate (deoxyguanosine triphosphate) to be incorporated into viral DNA, thereby terminating DNA synthesis.
The drug is intended for long-term use and significantly reduces viral load, helping to slow the progression of liver fibrosis and cirrhosis.
Indications
Entecavir is indicated for the treatment of chronic hepatitis B virus (HBV) infection in adults and pediatric patients (aged 2 years and older):
- Active Viral Replication: evidence of detectable virus in the blood and elevated liver enzyme levels (ALT/AST).
- Histological Evidence: presence of active inflammation or liver fibrosis confirmed by biopsy or non-invasive methods.
- Decompensated Liver Disease: used in treatment protocols for severe liver damage under close clinical supervision.
Dosage and administration
Entecavir must be taken at a consistent time to ensure maximum therapeutic efficacy.
- Standard Adult Dose: 0.5 mg once daily.
- Lamivudine-Refractory Patients: the dose is increased to 1 mg once daily.
- Administration Rules: must be taken on an empty stomach (at least 2 hours before or 2 hours after a meal).
- Renal Impairment: dosage adjustment or interval modification is mandatory for patients with a creatinine clearance below 50 mL/min.