Lapatinib – Targeted Cancer Therapy
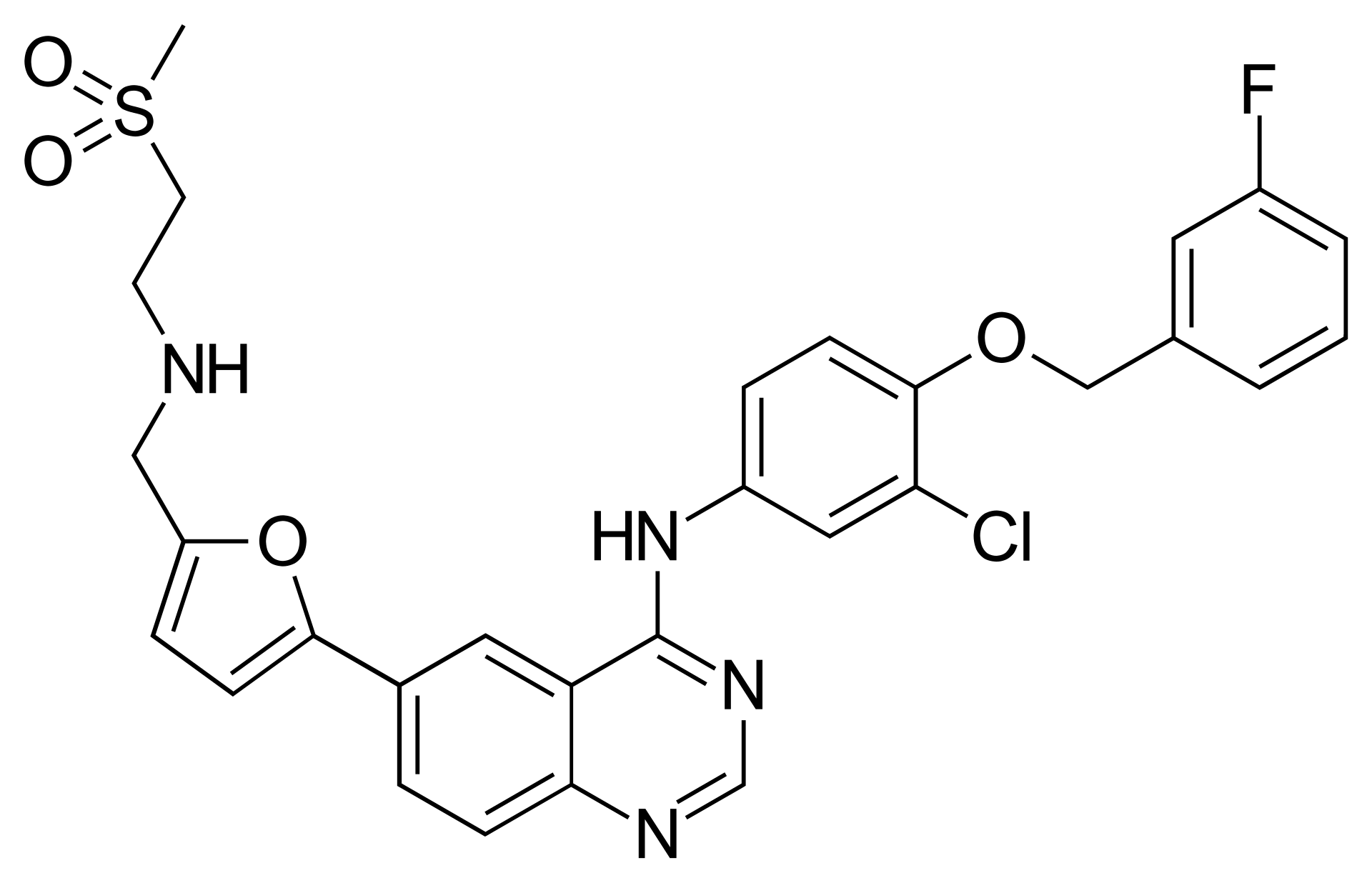
Lapatinib is a potent targeted anti-cancer medication belonging to the group of tyrosine kinase inhibitors (TKIs). It is a unique small-molecule compound capable of simultaneously targeting two key receptors responsible for cancer cell growth and division.
The mechanism of action involves the reversible inhibition of the intracellular tyrosine kinase domains of both EGFR (ErbB1) and HER2 (ErbB2) receptors. Unlike monoclonal antibodies that bind to the extracellular portion of the receptor, lapatinib penetrates the cell to block signaling from within. This results in cell cycle arrest and apoptosis of tumor cells that overexpress HER2. This dual inhibition provides a more effective suppression of tumor growth and helps overcome resistance to therapies that target only one receptor type.
Administered orally, lapatinib is frequently used in combination with other chemotherapeutic agents or endocrine therapy.
Indications
Lapatinib is indicated for the treatment of adult patients in the following clinical scenarios:
- HER2-positive Breast Cancer: advanced or metastatic breast cancer overexpressing HER2, in combination with capecitabine (following progression on trastuzumab).
- Hormone Receptor-Positive Breast Cancer: in combination with an aromatase inhibitor (e.g., letrozole) for postmenopausal women with metastatic disease.
- Combination Therapy: may also be used in combination with trastuzumab for patients with progressive disease.
Dosage and administration
Lapatinib dosage depends on the specific treatment regimen and concomitant medications.
- With Capecitabine: the standard dose is 1250 mg (five tablets) taken once daily continuously.
- With Letrozole: the standard dose is 1500 mg (six tablets) taken once daily.
- Administration Rules: must be taken at least 1 hour before or 1 hour after a meal. The full daily dose should be taken at once.
- Restrictions: avoid grapefruit juice, as it significantly increases the drug’s concentration in the blood.
- Dose Modification: if severe toxicity occurs (e.g., diarrhea or rash), the dose may be temporarily reduced or interrupted.