Larotrectinib: TRK Inhibitor for NTRK Fusion Cancers
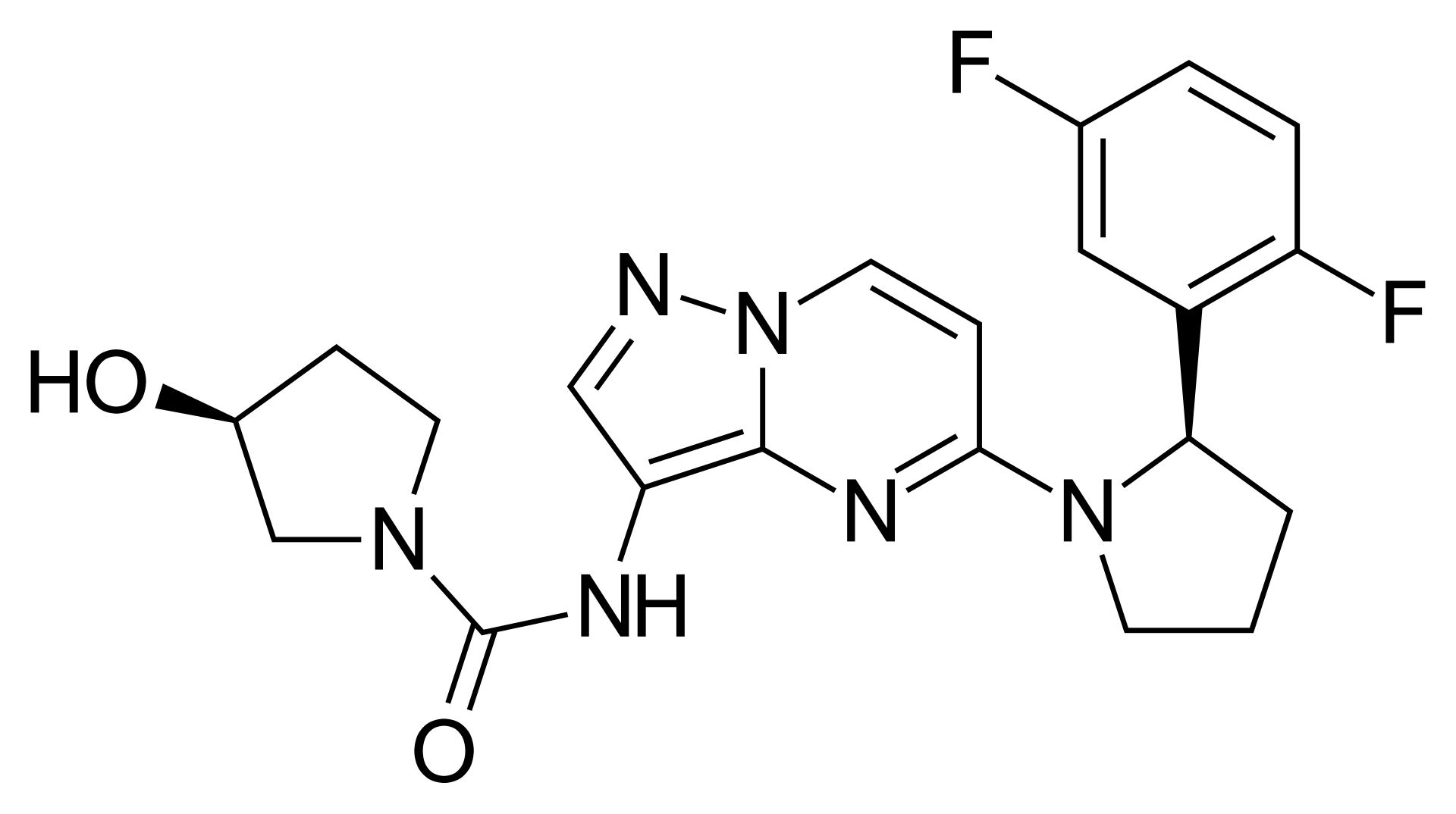
Larotrectinib is a first-in-class, highly selective tropomyosin receptor kinase (TRK) inhibitor. It is a "tissue-agnostic" cancer therapy, meaning it treats cancer based on a specific genetic mutation rather than where the cancer originated in the body.
The drug targets tumors with an NTRK gene fusion, blocking the abnormal TRK proteins that drive cancer cell growth. On Unifarm, you can find details regarding original medications (such as Vitrakvi) containing this active ingredient.
Indications
- Solid Tumors (Tumor-Agnostic): Treatment of adult and pediatric patients with solid tumors that have a Neurotrophic Tyrosine Receptor Kinase (NTRK) gene fusion without a known acquired resistance mutation.
- Specific Conditions:
- The disease is metastatic or where surgical resection is likely to result in severe morbidity.
- There are no satisfactory alternative treatments or the cancer has progressed following treatment.
Dosage and administration
Available as capsules and oral solution. Taken orally twice daily.
Standard Regimen:
- Adults: 100 mg twice daily.
- Pediatric Patients: 100 mg/m² body surface area twice daily (up to a maximum of 100 mg per dose).
Capsules should be swallowed whole. Do not chew or crush. Dosage modifications may be required based on toxicity or concomitant use of strong CYP3A4 inhibitors.