Mitomycin – Targeted Therapy
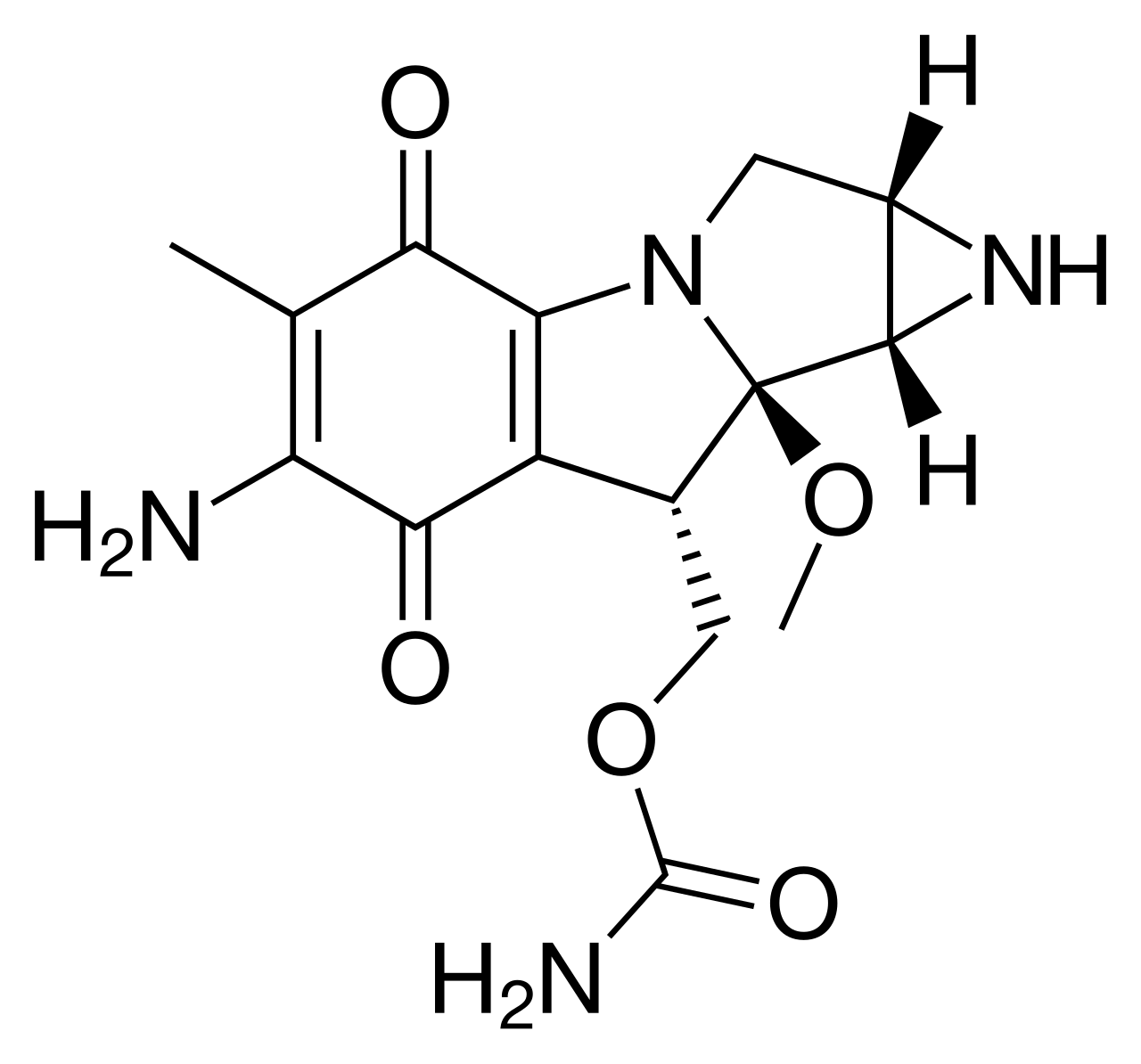
Mitomycin, also known as Mitomycin-C, is an antineoplastic antibiotic isolated from the culture of Streptomyces caespitosus. The drug acts as a bifunctional alkylating agent that causes cross-linking in the DNA molecule. It possesses phase-nonspecific activity, inhibiting DNA synthesis and, to a lesser extent, RNA and protein synthesis in rapidly dividing cells.
The mechanism of action of mitomycin involves its intracellular activation through enzymatic reduction. The activated form of the drug creates strong covalent bonds between complementary DNA strands, making their separation and replication impossible. This leads to chromosomal fragmentation and tumor cell death. Due to its ability to selectively suppress the growth of tissues with high metabolic rates, mitomycin is effective against various forms of adenocarcinomas.
The drug is administered intravenously or topically (intravesically for bladder cancer). It is rapidly distributed in tissues and metabolized primarily in the liver. A key feature of mitomycin is its cumulative myelotoxicity, which requires careful planning of intervals between treatment cycles.
Indications
Mitomycin is used in oncological practice both as monotherapy and as part of comprehensive treatment regimens:
- Gastrointestinal Cancers: adenocarcinoma of the stomach and pancreas (in combination with fluorouracil and doxorubicin).
- Bladder Cancer: intravesical therapy for treating superficial tumors and preventing recurrence after resection.
- Head and Neck Tumors: comprehensive treatment of squamous cell carcinoma in combination with radiation therapy.
- Lung Cancer: as part of combined chemotherapy regimens for non-small cell lung cancer.
- Cervical and Anal Canal Cancer: systemic treatment of advanced forms of the disease.
Dosage and administration
Mitomycin dosing schedules depend on the route of administration and the treatment protocol.
- Intravenous Administration: the standard dose is 10–20 mg/m² of body surface area every 6–8 weeks. Long intervals are necessary due to delayed toxicity to the bone marrow.
- Intravesical Administration: typically 20–40 mg of the drug dissolved in saline, administered directly into the bladder once a week for 6–8 weeks.
- Solution Preparation: the drug should be dissolved immediately before administration, avoiding skin contact (it is a potent irritant).
- Precautions: extravasation (leakage under the skin) must be strictly avoided, as mitomycin causes severe soft tissue necrosis.
- Blood Monitoring: monitoring of hematological parameters should be conducted weekly throughout the entire period between cycles.