Olaparib – PARP Inhibitor Therapy
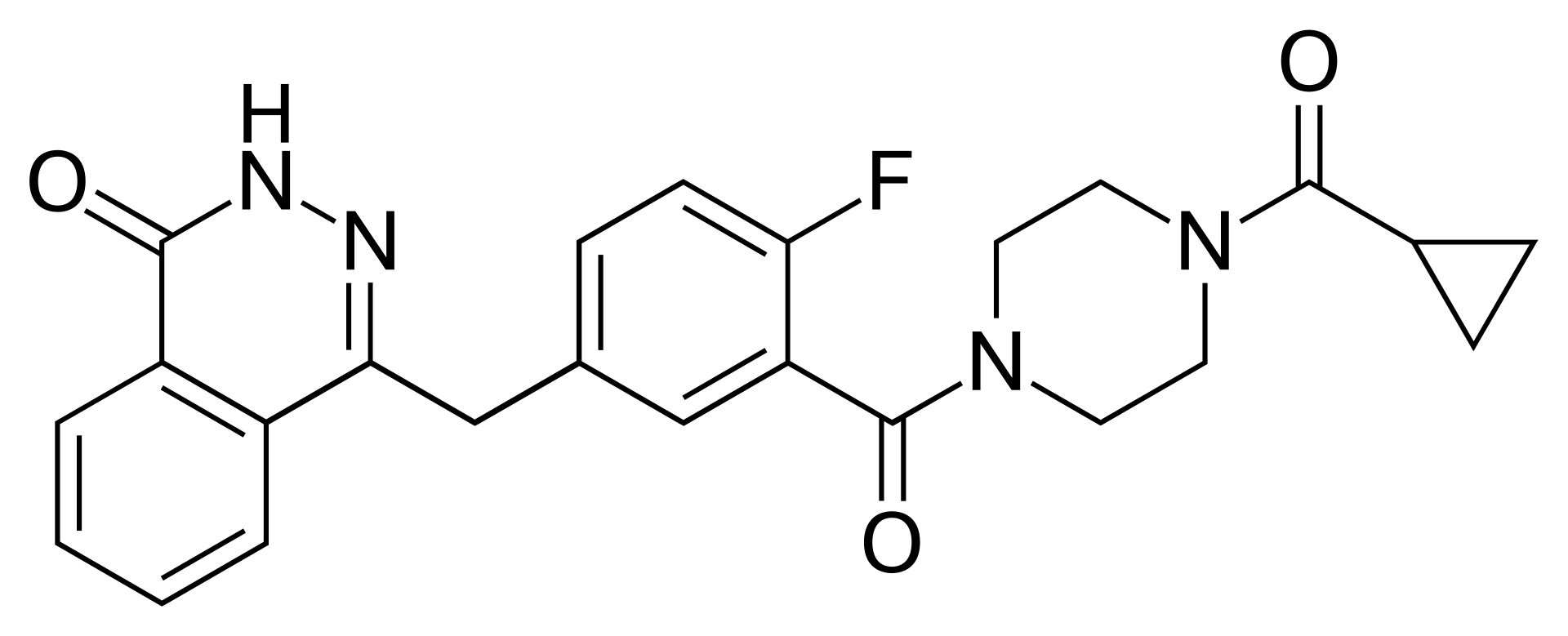
Olaparib is an innovative anti-tumor agent and the first-in-class inhibitor of poly(ADP-ribose) polymerase (PARP) enzymes. The drug represents a breakthrough in the concept of "synthetic lethality," selectively eliminating cancer cells with DNA repair deficiencies.
The mechanism of action involves blocking PARP1 and PARP2 enzymes, which are critical for repairing single-strand DNA breaks. When olaparib inhibits these enzymes, single-strand breaks are converted into more lethal double-strand breaks during replication. Healthy cells repair such damage through homologous recombination. However, in tumor cells with BRCA1 or BRCA2 mutations, this repair pathway is dysfunctional. Consequently, the accumulation of unrepaired DNA damage leads specifically to the death of cancer cells while sparing healthy tissues to a greater extent.
The medication is available in oral tablet form and is used for both maintenance therapy and the treatment of advanced malignancies.
Indications
Olaparib is indicated for the treatment of adult patients with the following types of malignancies:
- Ovarian Cancer: maintenance treatment of recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer.
- Breast Cancer: treatment of HER2-negative metastatic breast cancer with germline BRCA mutations.
- Pancreatic Cancer: maintenance treatment of metastatic pancreatic adenocarcinoma with germline BRCA mutations.
- Prostate Cancer: treatment of metastatic castration-resistant prostate cancer (mCRPC) with mutations in DNA repair genes.
Dosage and administration
Olaparib dosing must be strictly followed to maintain therapeutic levels in the blood.
- Standard Dose: 300 mg (two 150 mg tablets) taken twice daily.
- Total Daily Dose: 600 mg.
- Administration: tablets should be swallowed whole, with or without food.
- Interactions: it is essential to avoid grapefruit juice and Seville oranges (pomelo), as they increase drug concentration to potentially toxic levels.
- Adjustment: in patients with moderate renal impairment, the dose is reduced to 200 mg twice daily.