Plerixafor – Targeted Therapy
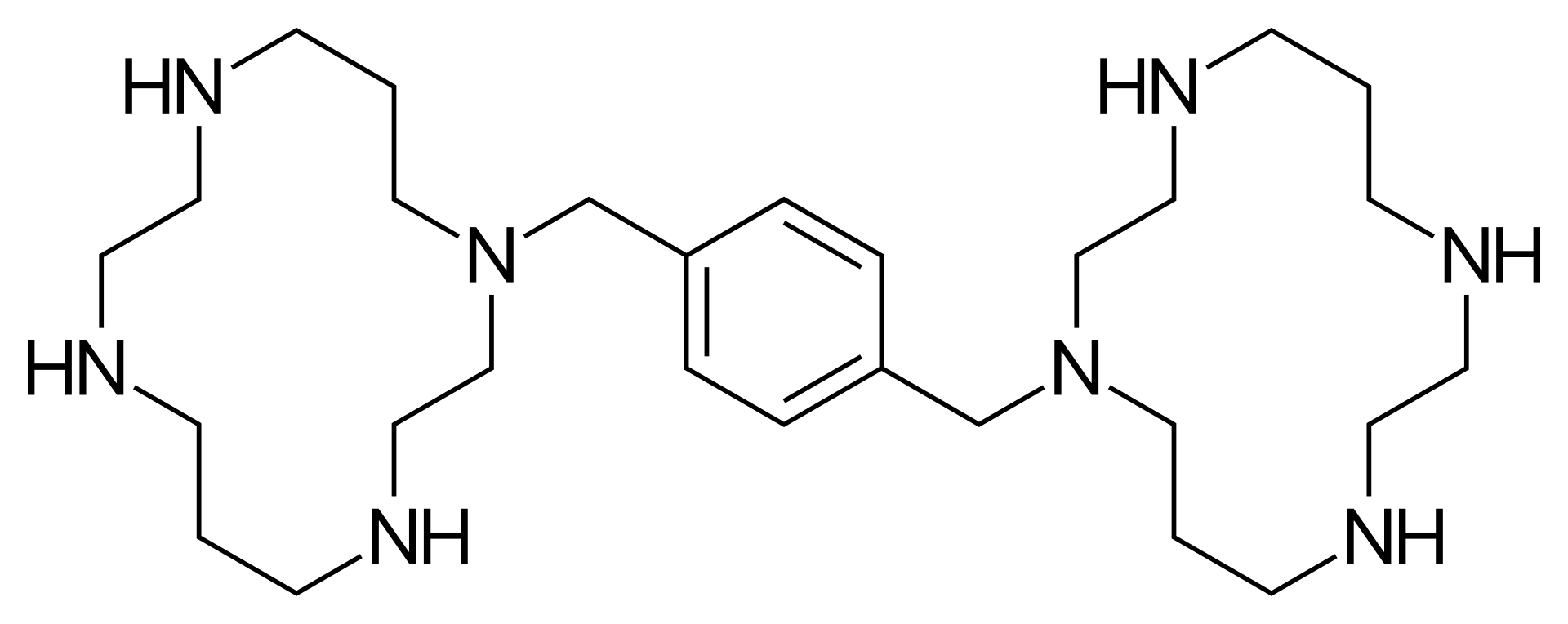
Plerixafor is an innovative hematopoietic stem cell mobilization stimulator, functioning as a selective CXCR4 receptor antagonist. The drug belongs to the bicyclam class and is used in transplantology to increase the egress of stem cells from the bone marrow into the systemic circulation for subsequent collection.
The mechanism of action of plerixafor involves blocking the binding of the chemokine SDF-1α (also known as CXCL12) to its receptor CXCR4. Normally, the interaction between SDF-1α and CXCR4 tethers hematopoietic stem cells within the bone marrow microenvironment. Plerixafor disrupts this bond, leading to the rapid release (mobilization) of stem cells into the peripheral blood. Peak cell concentrations in the blood are typically reached 10–14 hours after administration.
The drug is administered subcutaneously and acts with high speed. Plerixafor significantly increases the likelihood of successfully collecting the required number of CD34+ cells, particularly in patients who previously responded poorly to standard growth factor mobilization.
Indications
Plerixafor is used in hematological practice for preparation for autologous transplantation:
- Multiple Myeloma: mobilization of hematopoietic stem cells into peripheral blood for collection and transplantation in adult patients.
- Lymphomas (Hodgkin’s and Non-Hodgkin’s): stimulation of cell egress in lymphoma patients to ensure sufficient graft volume.
- Combination Therapy: the drug is always used in conjunction with granulocyte colony-stimulating factor (G-CSF).
- Pediatric Hematological Diseases: mobilization of cells in children with lymphomas or solid tumors when autologous transplantation is required.
Dosage and administration
The plerixafor administration regimen is strictly timed to the apheresis schedule (the cell collection procedure).
- Standard Dose: 240 mcg per kg of body weight (for weight ≤83 kg, a fixed dose of 20 mg may be used).
- Route of Administration: the drug is administered subcutaneously approximately 11 hours before the start of the apheresis session.
- Course of Treatment: injections are given daily for up to 4 consecutive days until the target number of stem cells is collected.
- Dose Adjustment: for patients with moderate or severe renal impairment, the dose is reduced to 160 mcg/kg.
- Concomitant Preparation: plerixafor is prescribed after a 4-day course of G-CSF, with G-CSF injections continuing on the days of plerixafor administration.