Remdesivir – Antiviral Therapy
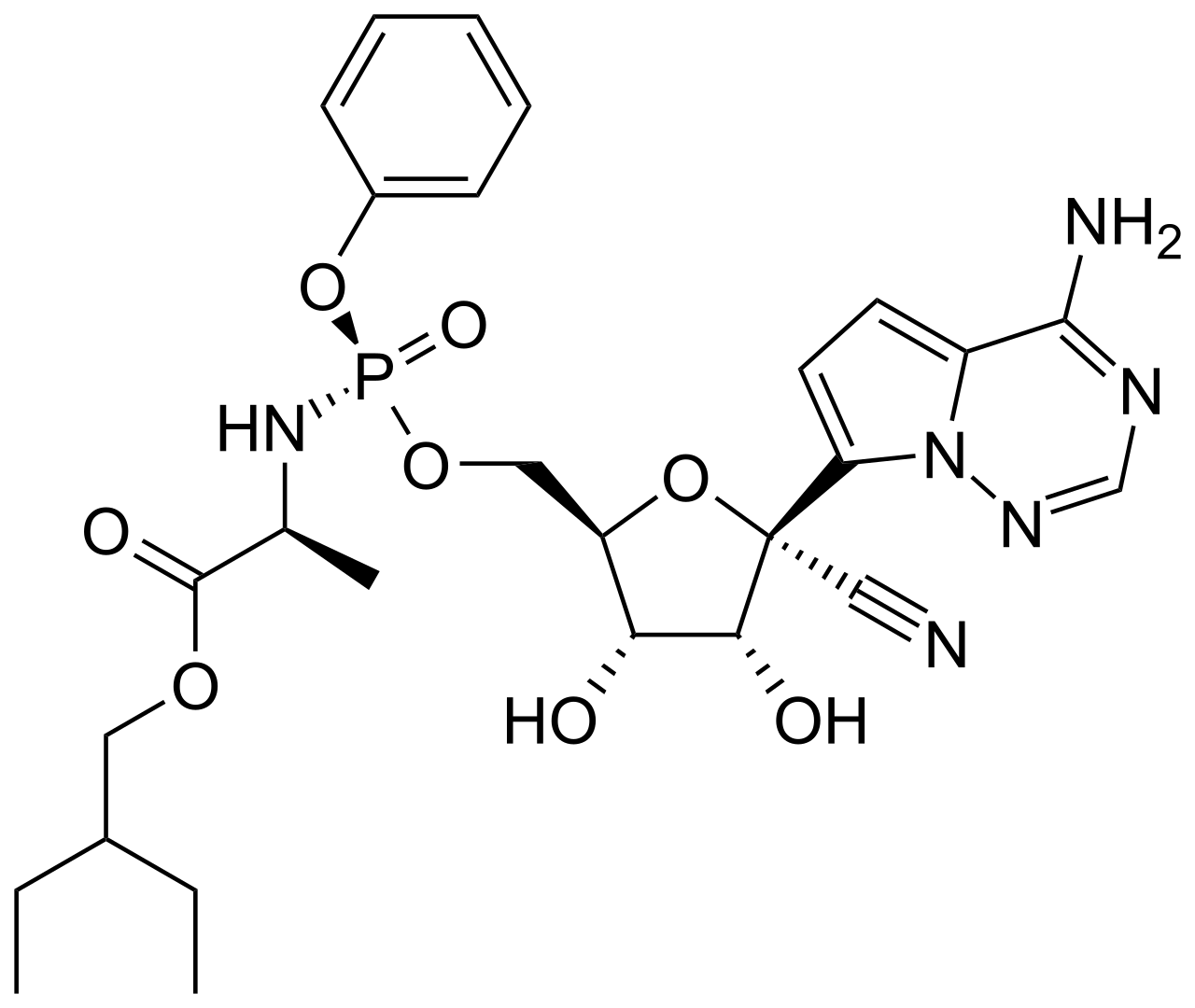
Remdesivir is a direct-acting antiviral medication and a nucleotide analog. Originally developed to target the Ebola virus, it gained global prominence as the first drug formally approved for the treatment of severe COVID-19 infections.
The mechanism of action involves the inhibition of viral RNA-dependent RNA polymerase. Functioning as a "false" building block (an analog of adenosine triphosphate), the drug is incorporated into the growing viral RNA strand. This leads to premature termination of RNA synthesis (delayed chain termination), effectively blocking viral replication within the host cells. Remdesivir exhibits broad-spectrum activity against various RNA viruses, including members of the Coronaviridae and Filoviridae families.
The drug is strictly intended for hospital use and is administered via intravenous infusion.
Indications
Remdesivir is indicated for the treatment of viral infections in adults and pediatric patients (weighing at least 3.5 kg):
- COVID-19: treatment of moderate to severe coronavirus disease in hospitalized patients requiring supplemental oxygen.
- High-Risk Patients: therapy for non-hospitalized individuals with confirmed COVID-19 who are at high risk of progressing to severe disease.
Dosage and administration
Remdesivir treatment courses typically last for 5 to 10 days, depending on the patient's clinical progression.
- Adults and Pediatrics (over 40 kg): a single loading dose of 200 mg on Day 1, followed by maintenance doses of 100 mg once daily.
- Pediatrics (3.5 kg to 40 kg): a loading dose of 5 mg/kg on Day 1, followed by maintenance doses of 2.5 mg/kg once daily.
- Administration: exclusively via intravenous infusion over a period of 30 to 120 minutes.
- Monitoring: renal and hepatic function tests (ALT, AST) must be performed before and during treatment.