Etoposide – Cancer & Lymphoma Treatment
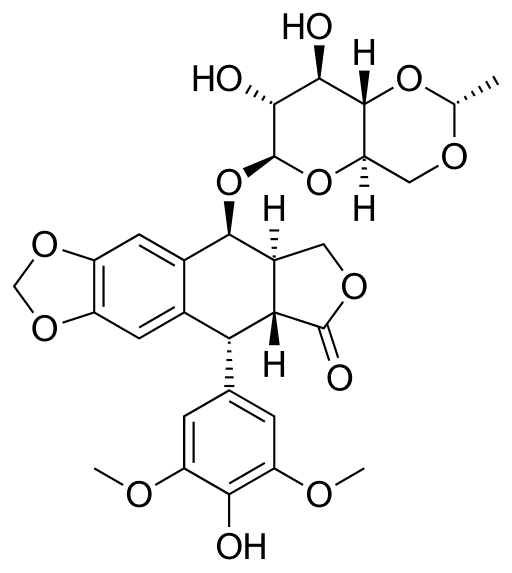
Etoposide is a semi-synthetic antineoplastic agent derived from podophyllotoxin. The drug belongs to the group of topoisomerase II inhibitors and is a phase-specific cytostatic that acts primarily in the S and G2 phases of the cell cycle. Etoposide is widely used in oncology due to its ability to effectively induce the death of rapidly dividing malignant cells.
The mechanism of action of etoposide involves forming a stable complex with the topoisomerase II enzyme and the DNA molecule. This prevents the re-ligation of breaks in the DNA double helix that occur during replication. The accumulation of such unrepairable damage blocks cell division and triggers programmed cell death (apoptosis). The drug is effective against a broad spectrum of solid tumors and hematologic malignancies.
Etoposide is available in formulations for intravenous administration and oral intake. It has a high capacity to penetrate various body tissues, although it crosses the blood-brain barrier in a limited volume. The drug is metabolized in the liver and excreted primarily by the kidneys.
Indications
Etoposide is a key component of many chemotherapy protocols and is prescribed for the following conditions:
- Germ Cell Tumors: testicular and ovarian cancer (often in combination with cisplatin and bleomycin).
- Lung Cancer: small cell lung cancer (SCLC) as first-line therapy.
- Hematologic Malignancies: acute myeloid leukemia, Hodgkin's lymphoma, and non-Hodgkin lymphomas.
- Pediatric Tumors: neuroblastoma, Ewing's sarcoma, and Wilms' tumor (as part of complex regimens).
- Other Solid Tumors: gastric cancer, breast cancer (in specific regimens when resistant to other therapy).
Dosage and administration
The dosing regimen of etoposide is established individually based on the diagnosis, the patient's body surface area (BSA), and renal function.
- Intravenous Administration: the standard dose is 50–120 mg/m² daily for 3–5 days every 3–4 weeks. The infusion must be administered slowly (at least 30–60 minutes).
- Oral Intake: the oral dose is typically twice the intravenous dose (due to approximately 50% bioavailability) and ranges from 100–200 mg/m² daily.
- Administration Specifics: rapid intravenous injection may lead to a sudden drop in blood pressure (hypotension).
- Dose Adjustment: required in cases of decreased creatinine clearance or significant liver function impairment.
- Monitoring: a complete blood count (especially leukocyte and platelet levels) must be monitored before each cycle.