Fosfestrol – Targeted Therapy
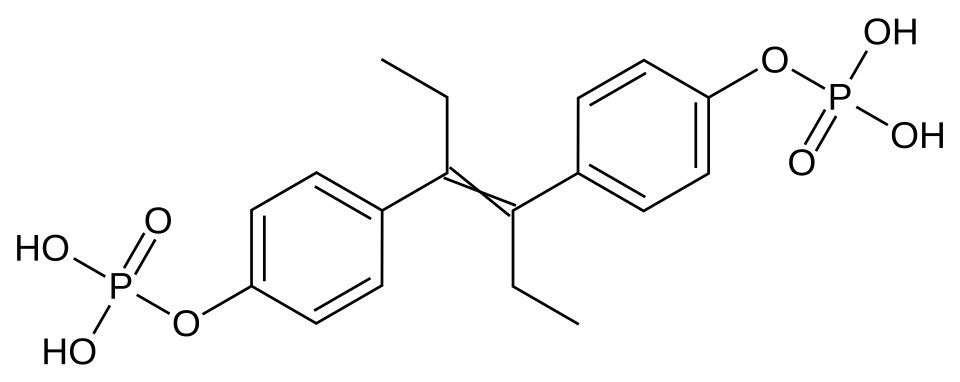
Fosfestrol is a synthetic estrogenic agent, chemically identified as diethylstilbestrol diphosphate. It is a "prodrug" specifically developed for the targeted therapy of hormone-dependent prostate neoplasms. The primary pharmacological value of fosfestrol lies in its ability to target tumor tissue while minimizing systemic estrogenic effects during the initial stages of metabolism.
The mechanism of action of fosfestrol is unique, utilizing the high activity of the acid phosphatase enzyme within prostate cancer cells. Following administration, the drug undergoes dephosphorylation directly within the tumor tissue and its metastases under the influence of this enzyme, releasing active diethylstilbestrol. This creates a high local concentration of estrogen that inhibits cancer cell division. Additionally, the drug exerts a powerful antiandrogenic effect by inhibiting the secretion of pituitary gonadotropic hormones, leading to a sharp decline in endogenous testosterone levels.
Fosfestrol also possesses direct cytotoxic activity against tumor cells that is independent of hormone receptors, allowing its use even when resistance to standard hormonal therapy has developed.
Indications
Fosfestrol is primarily used in onco-urological practice for the treatment of specific conditions:
- Prostate Cancer: therapy for progressive, locally advanced, or metastatic prostate cancer (especially in late stages).
- Hormone-Refractory Prostate Cancer: treatment of patients for whom standard androgen blockade or surgical castration has ceased to provide a therapeutic effect.
- Tumor Recurrence: management of recurrent forms of the disease following radiation therapy or prostatectomy.
- Bone Metastases: reduction of pain and slowing the growth of metastatic foci in bone tissue associated with prostate cancer.
Dosage and administration
The fosfestrol treatment regimen is individually tailored based on disease severity and the method of administration (intravenous or oral).
- Intravenous Administration (Intensive Phase): typically begins with 600–1200 mg per day via slow infusion for the first 5–10 days to achieve a rapid effect.
- Maintenance Therapy: after symptom reduction, the dose is gradually decreased to 300 mg 1–3 times weekly intravenously, or transitioned to oral tablet intake.
- Oral Intake: the initial dose is 100–200 mg three times daily, followed by individual selection of a maintenance dose.
- Administration Specifics: intravenous injections must be performed very slowly (over 5–10 minutes) under medical supervision due to the risk of acute vascular reactions.
- Duration of Treatment: therapy is usually long-term, with regular monitoring of prostate-specific antigen (PSA) levels and acid phosphatase activity.