Valganciclovir – Antiviral Therapy
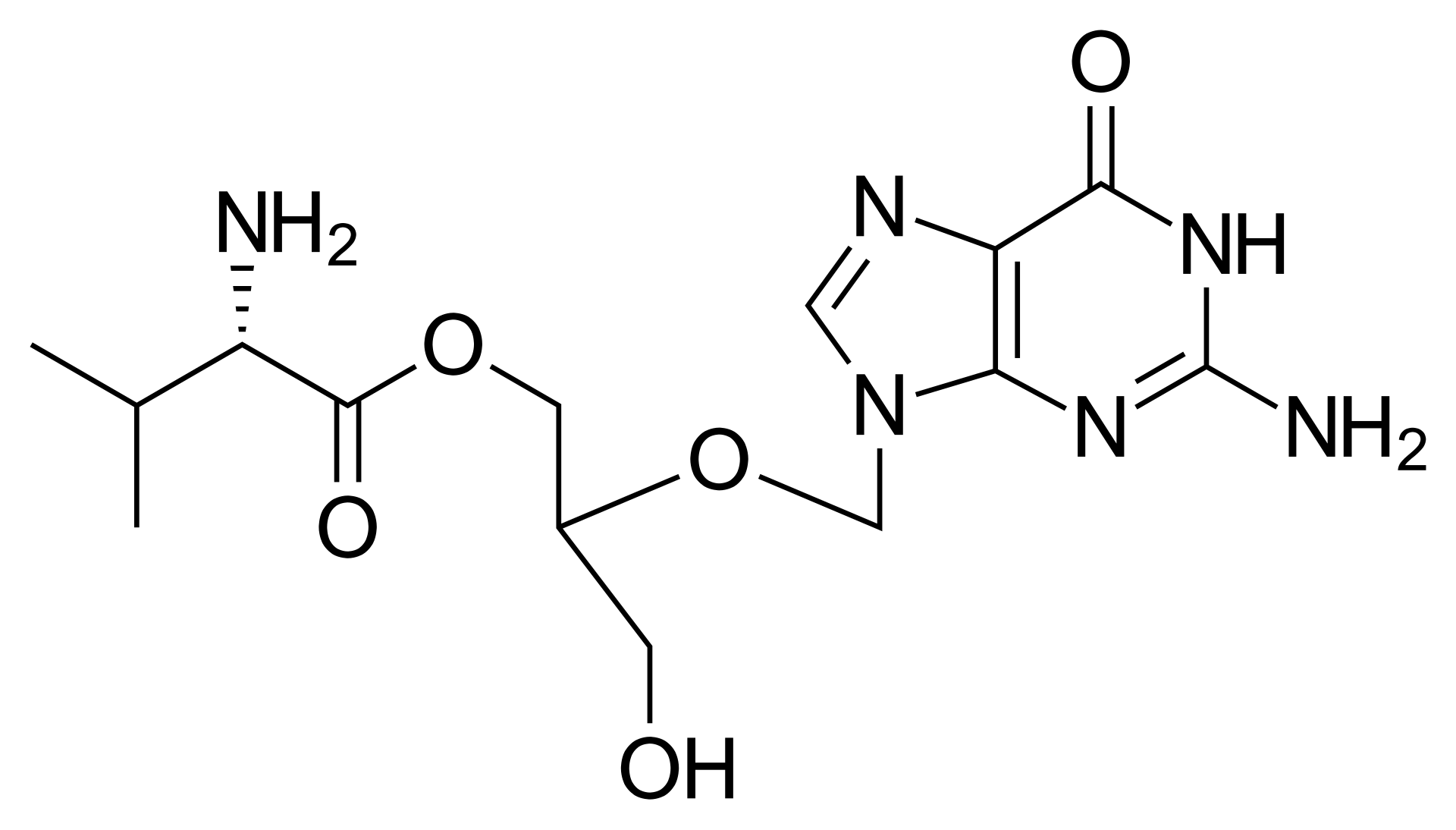
Valganciclovir is an antiviral medication that acts as an L-valyl ester prodrug of ganciclovir. It was developed to provide high oral bioavailability, making it an effective option for managing severe herpesvirus infections.
The mechanism of action involves the rapid conversion of valganciclovir into ganciclovir by intestinal and hepatic esterases. Inside virus-infected cells, ganciclovir is phosphorylated into its active triphosphate form. This compound competitively inhibits viral DNA polymerase and is incorporated into viral DNA, resulting in the termination of viral DNA synthesis. Valganciclovir exhibits potent activity against cytomegalovirus (CMV), herpes simplex virus types 1 and 2, human herpesvirus types 6, 7, and 8, and Epstein-Barr virus.
The drug is a primary choice for CMV prophylaxis in organ transplant recipients and for the treatment of CMV retinitis in immunocompromised patients.
Indications
Valganciclovir is indicated for the treatment of adult and pediatric patients in the following cases:
- CMV Retinitis: treatment of cytomegalovirus retinitis in patients with acquired immunodeficiency syndrome (AIDS).
- CMV Prophylaxis: prevention of CMV disease in solid organ transplant recipients (kidney, heart, pancreas) at high risk for infection.
Dosage and administration
Valganciclovir dosing must be strictly followed due to the risk of toxicity.
- CMV Retinitis (Induction): 900 mg twice daily for 21 days.
- Maintenance and Prophylaxis: 900 mg once daily.
- Administration Rules: tablets should be taken with food to maximize absorption.
- Renal Impairment: careful dose adjustment is required based on creatinine clearance.
- Special Handling: tablets should not be broken or crushed due to potential teratogenic and carcinogenic risks.