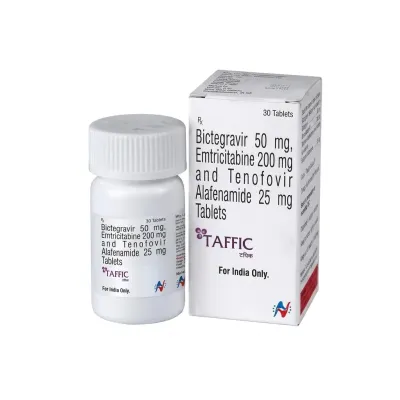
Bictegravir – HIV Antiviral Therapy
Bictegravir is an innovative second-generation integrase strand transfer inhibitor (INSTI) with high antiviral potency and a high genetic barrier to resistance. The drug is a cornerstone component of modern HIV treatment regimens.
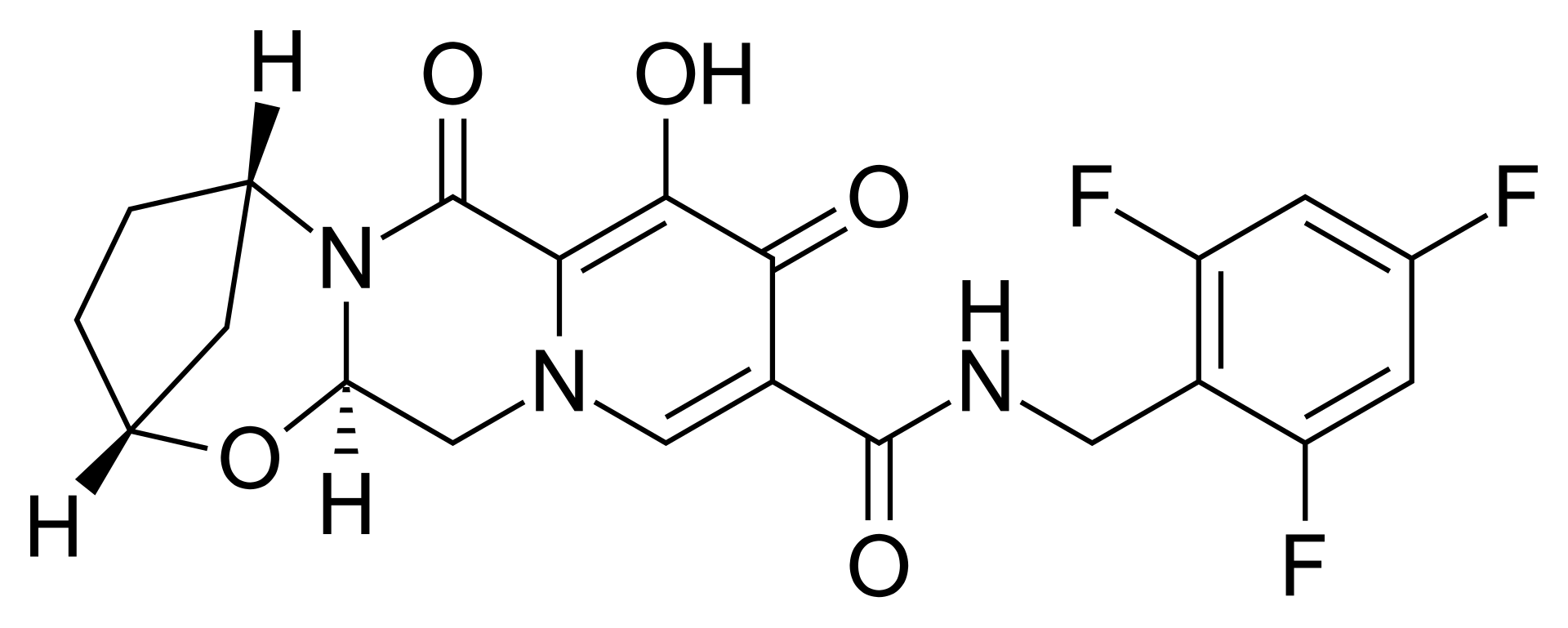
The mechanism of action of bictegravir involves blocking integrase, an enzyme required by HIV to incorporate its viral DNA into the host cell's (CD4+ T-lymphocyte) genetic material. The drug binds tightly to the active site of the integrase, making the integration process impossible and effectively stopping viral replication at an early stage. Due to its chemical structure, bictegravir remains effective even against virus strains that have developed resistance to first-generation integrase inhibitors.
The drug is characterized by excellent tolerability and a long half-life, allowing for once-daily dosing. It is typically administered as part of "three-in-one" combination tablets.
Indications
Bictegravir is indicated for the treatment of HIV-1 infection in adults and pediatric patients weighing at least 14 kg:
- Naive Patients: initial therapy for patients who have not previously received antiretroviral therapy (ART).
- Stable Patients: as a replacement for current regimens in patients with achieved virologic suppression (HIV-1 RNA < 50 copies/mL) and no known resistance to integrase inhibitors.
Dosage and administration
Bictegravir dosing is standardized and generally does not require complex adjustment for most patients.
- Standard Adult Dose: 50 mg once daily.
- Administration Rules: the drug is taken orally, with or without food.
- Missed Dose: if less than 18 hours have passed since the missed dose, it should be taken as soon as possible; if more than 18 hours, skip to the next scheduled dose.
- Drug Interactions: should be taken at least 2 hours before or 6 hours after antacids or supplements containing magnesium, aluminum, iron, or calcium.