Emtricitabine – Antiviral HIV Therapy
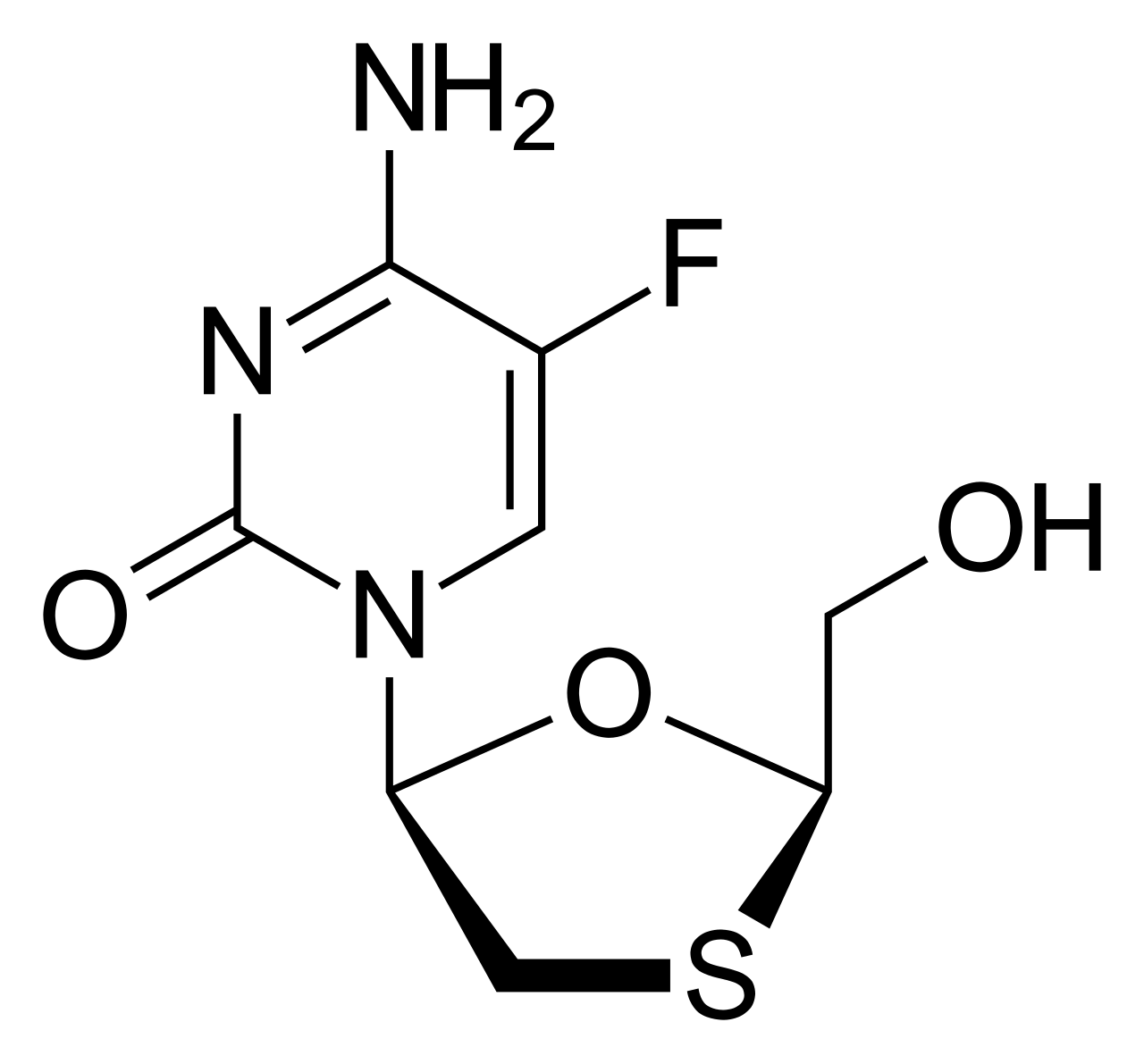
Emtricitabine is a potent nucleoside reverse transcriptase inhibitor (NRTI), a cytidine analogue used for the treatment of HIV infection and Hepatitis B virus. The drug is a cornerstone component of modern antiretroviral therapy.
The mechanism of action of emtricitabine involves its intracellular phosphorylation into its active triphosphate form. In this state, it competes with the natural substrate (deoxycytidine 5'-triphosphate) and is incorporated into the developing viral DNA chain. This results in immediate chain termination, as the lack of the necessary chemical bond blocks further replication of the virus's genetic material. Due to its high specificity for viral enzymes, the drug effectively suppresses the replication of HIV-1, HIV-2, and Hepatitis B virus (HBV), while maintaining low cytotoxicity.
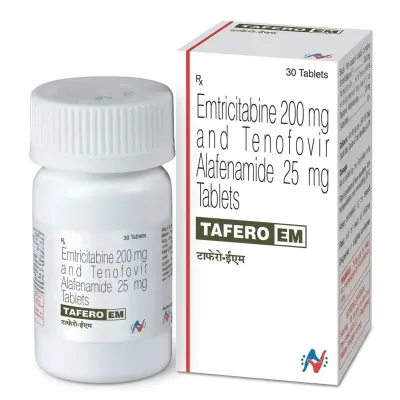
The drug is characterized by a long intracellular half-life, allowing for once-daily dosing. Emtricitabine is frequently used as part of fixed-dose combinations (e.g., with tenofovir).
Indications
Emtricitabine is used for the treatment of adult and pediatric patients in the following cases:
- HIV-1 Infection: as part of combination antiretroviral therapy to control viral load.
- Pre-exposure Prophylaxis (PrEP): in combination with other agents to reduce the risk of HIV-1 acquisition in high-risk individuals.
- Chronic Hepatitis B: as an additional benefit when treating co-infected HIV patients (the drug is active against HBV).
Dosage and administration
Emtricitabine dosing is standardized but requires adjustment in patients with impaired renal function.
- Standard Adult Dose: 200 mg once daily.
- Administration Rules: capsules are taken orally regardless of food intake.
- Pediatric Dosage: calculated individually based on body weight (usually 6 mg/kg in solution or capsule form).
- Renal Impairment: for creatinine clearance less than 50 mL/min, the dosing interval must be increased or the dose reduced.
- Special Note: the drug should not be abruptly discontinued in patients co-infected with Hepatitis B due to the risk of severe disease flare-ups.
Frequently Asked Questions
List of medicines by active substance Emtricitabine