Oxaliplatin – Targeted Therapy
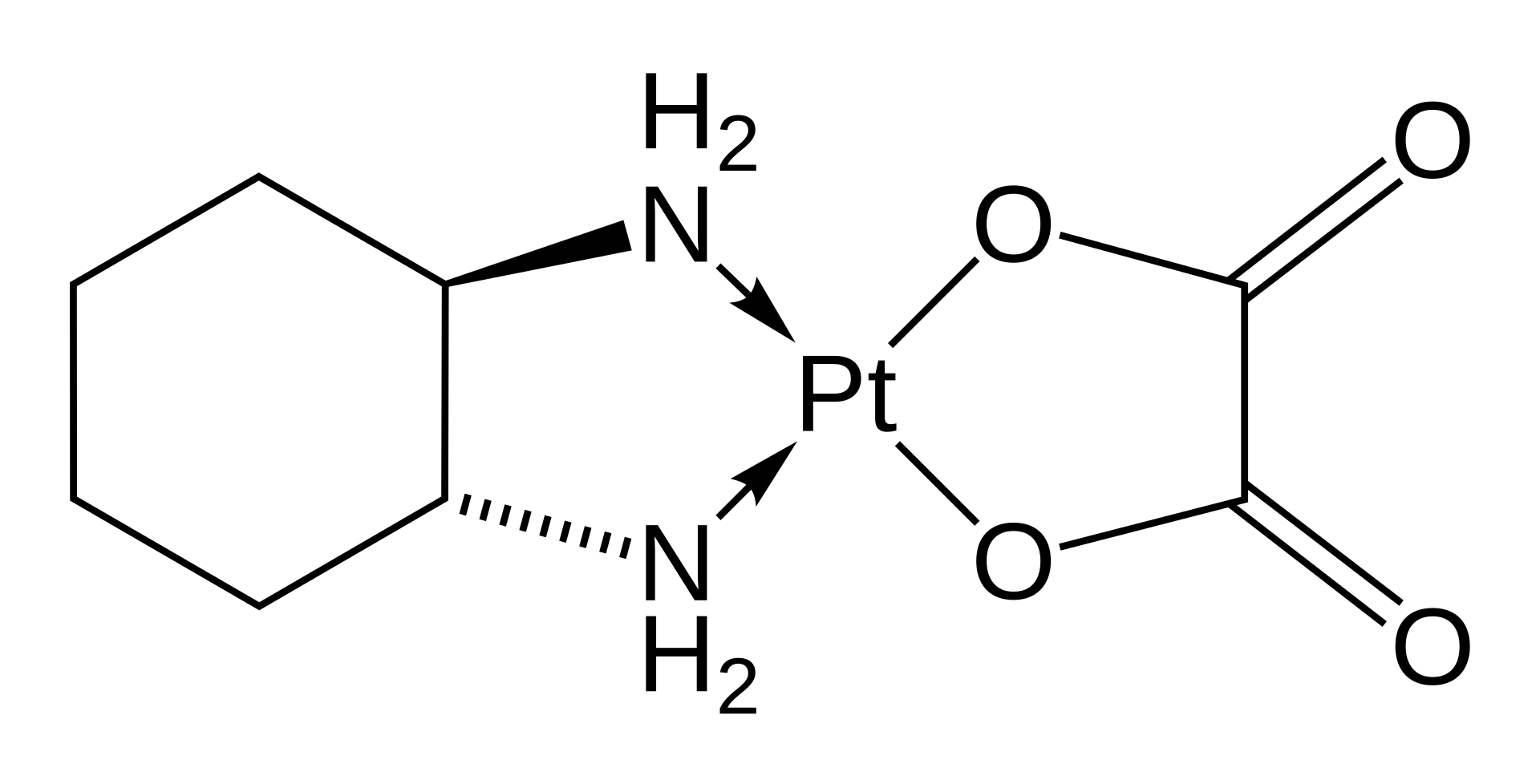
Oxaliplatin is an antineoplastic agent representing the third generation of platinum-based organic compounds. The drug belongs to the class of alkylating agents and is widely used in modern oncology due to its high cytotoxic activity and a more favorable safety profile compared to cisplatin.
The mechanism of action of oxaliplatin is based on its ability to interact with the DNA of tumor cells. Active derivatives of the drug form intrastrand and interstrand cross-links in the DNA molecule, which block replication and transcription processes. This leads to DNA strand breaks and subsequent malignant cell death via apoptosis. Oxaliplatin is effective even against tumor lines that have developed resistance to other platinum agents.
The drug is administered exclusively intravenously via infusion. Oxaliplatin exhibits significant synergism with fluoropyrimidines, making it a key component of standard chemotherapy protocols for colorectal cancer.
Indications
Oxaliplatin is indicated for the systemic treatment of malignant neoplasms of the gastrointestinal tract:
- Colorectal Cancer: adjuvant therapy of colon cancer following surgical resection of the primary tumor (Stage III).
- Metastatic Cancer: treatment of advanced colorectal cancer (usually in combination with 5-fluorouracil and leucovorin — FOLFOX regimens).
- Gastric Cancer: therapy for advanced forms of gastric adenocarcinoma and gastroesophageal junction cancer.
- Pancreatic Cancer: used as part of combination regimens (e.g., FOLFIRINOX) for metastatic disease.
Dosage and administration
The oxaliplatin dosing regimen is established individually based on body surface area and the specific treatment schedule.
- Standard Dose: 85 mg/m² (in FOLFOX regimens every 2 weeks) or 130 mg/m² (in XELOX regimens every 3 weeks).
- Route of Administration: intravenous infusion lasting from 2 to 6 hours. The drug must not be administered simultaneously with alkaline solutions or medications containing chlorides.
- Diluent: only 5% glucose (dextrose) solution is used to prepare the infusion solution.
- Dose Adjustment: if neurotoxicity or significant hematologic toxicity develops, the dose is reduced or the intervals between administrations are increased.
- Precautions: during and immediately after administration, the patient should avoid contact with cold objects and consumption of cold food/drinks.