Sunitinib – Targeted Therapy
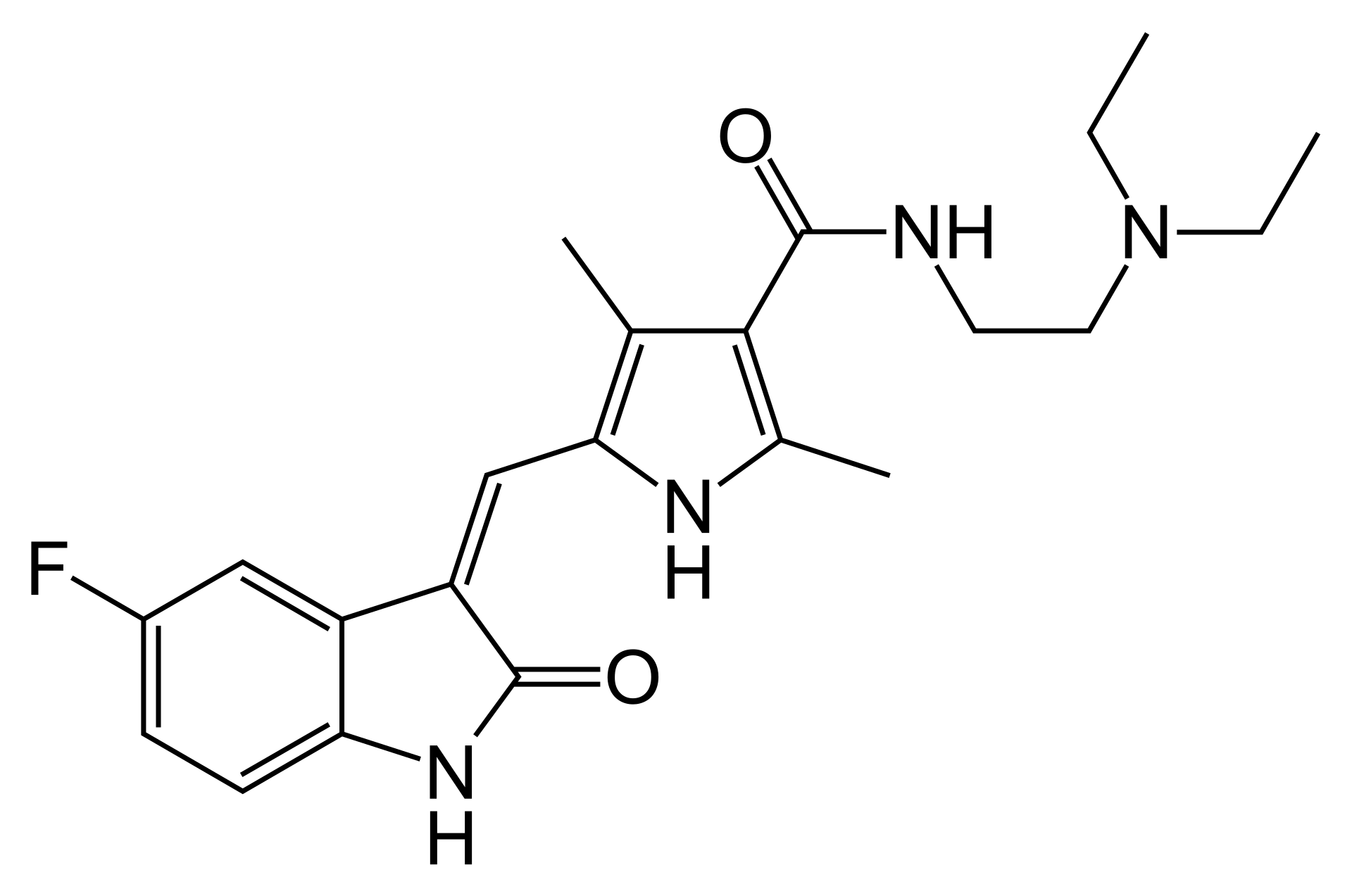
Sunitinib is a new-generation oral multi-kinase inhibitor with potent antitumor and antiangiogenic properties. The drug is designed to combat advanced forms of cancer that actively form their own network of blood vessels for growth.
The mechanism of action of sunitinib involves the selective blocking of tyrosine kinase receptors, including vascular endothelial growth factor receptors (VEGFR-1, -2, -3), platelet-derived growth factor receptors (PDGFR-α, -β), stem cell factor receptor (KIT), and Fms-like tyrosine kinase-3 (FLT3). This broad spectrum allows the drug to simultaneously "starve" the tumor by cutting off its blood supply and directly inhibit signals that cause cancer cells to divide uncontrollably. Sunitinib has demonstrated high efficacy in treating tumors resistant to standard chemotherapy.
The drug is administered orally as capsules. Its use significantly slows disease progression and increases survival rates in patients with severe oncological conditions.
Indications
Sunitinib is indicated for the treatment of adult patients in the following clinical cases:
- Gastrointestinal Stromal Tumors (GIST): following disease progression or intolerance to imatinib.
- Advanced and/or Metastatic Renal Cell Carcinoma (mRCC): as first-line therapy or following cytokine therapy failure.
- Pancreatic Neuroendocrine Tumors (pNET): progressive, well-differentiated tumors in patients with unresectable disease.
Dosage and administration
The dosage of sunitinib depends on the tumor type and the patient's drug tolerance.
- For GIST and RCC: the standard dose is 50 mg once daily for 4 weeks, followed by a 2-week break (4/2 schedule).
- For pNET: the recommended dose is 37.5 mg once daily, continuously, without a scheduled break.
- Administration Rules: capsules are taken orally, regardless of food intake, at the same time each day.
- Dose Adjustment: the physician may modify the dosage in 12.5 mg increments (minimum 25 mg, maximum 75 mg) based on the clinical response.
- Monitoring: regular monitoring of left ventricular ejection fraction and blood pressure is mandatory during treatment.