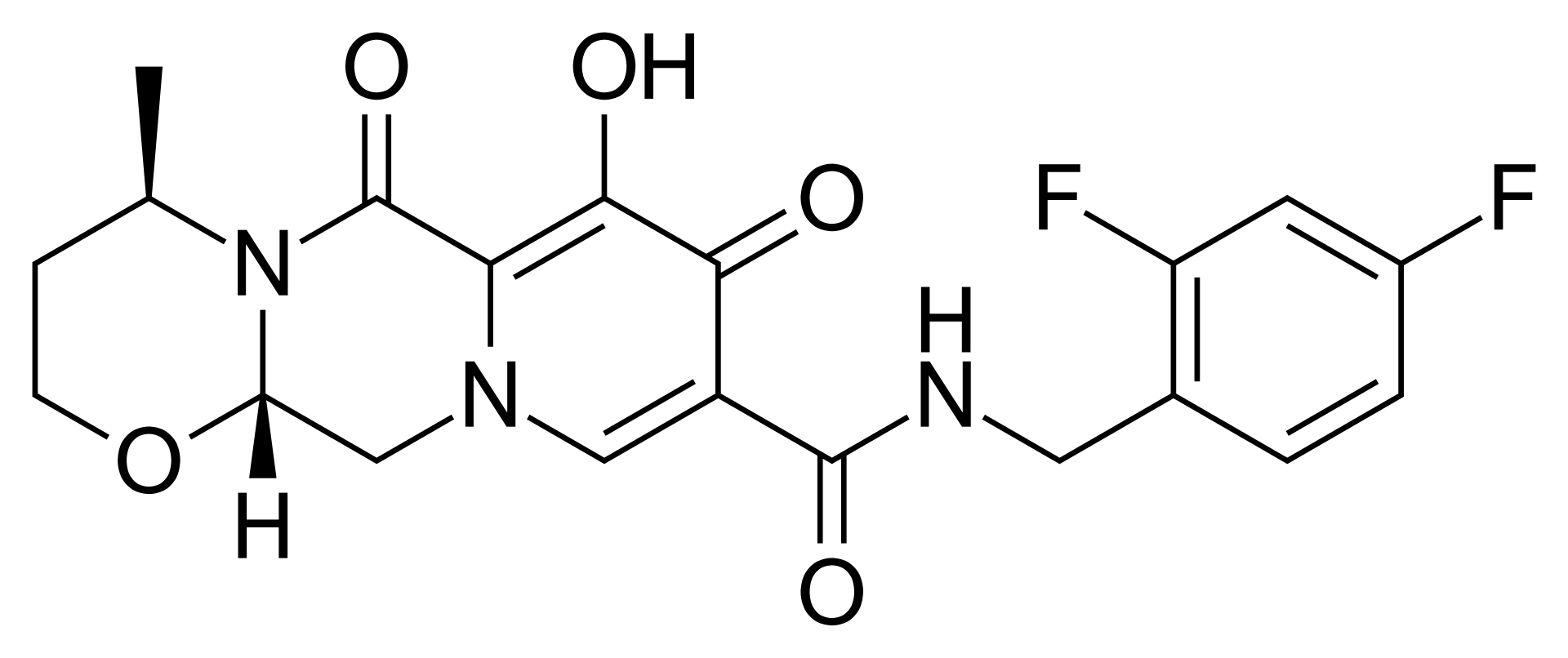
Dolutegravir – Antiretroviral Therapy
Dolutegravir is a second-generation antiretroviral agent belonging to the class of HIV-1 integrase strand transfer inhibitors (INSTIs). The drug is one of the most modern and effective components of highly active antiretroviral therapy (HAART), characterized by a high genetic barrier to the development of resistance and a favorable safety profile.
The mechanism of action of dolutegravir involves blocking the integrase enzyme, which is essential for the human immunodeficiency virus to incorporate viral DNA into the genetic apparatus of the host cell (CD4+ T-lymphocyte). The drug binds to the enzyme's active site and prevents the strand transfer step, thereby completely interrupting the viral replication cycle. This leads to a rapid and sustained reduction of plasma viral load to undetectable levels.
Dolutegravir is characterized by a long half-life, allowing for once-daily dosing. Due to its structural design, it maintains activity against HIV strains resistant to first-generation integrase inhibitors (such as raltegravir), making it an indispensable tool for treating both treatment-naive and treatment-experienced patients.
Indications
Dolutegravir is used as part of combination antiretroviral therapy for the treatment of HIV infection:
- HIV-1 infection in adults: treatment of patients who have not previously received therapy (naive) and treatment-experienced patients.
- HIV-1 infection in adolescents and children: approved for use in children weighing at least 3 kg (in specific dosage forms).
- Resistant forms of HIV: therapy for patients with identified partial resistance to other classes of antiretroviral drugs.
- Post-exposure prophylaxis (PEP): can be used in emergency prophylaxis regimens following potential exposure to HIV.
Dosage and administration
The dolutegravir administration regimen is standardized, though dosage may be adjusted based on the patient's treatment history.
- Standard Dosage: 50 mg (one tablet) once daily for patients without prior exposure to integrase inhibitors.
- In Case of Resistance: for patients with confirmed or suspected resistance to INSTIs, the dose is increased to 50 mg twice daily.
- Administration Rules: the drug can be taken with or without food. However, when co-administered with etravirine, dolutegravir must be taken with food.
- Drug Interactions: medications containing cations (magnesium/aluminum antacids, calcium, iron, multivitamins) should be taken 2 hours before or 6 hours after dolutegravir, as they reduce its absorption.
- Missed Dose: if more than 12 hours remain until the next dose, the missed dose should be taken immediately. If less than 12 hours remain, the missed dose should be skipped.