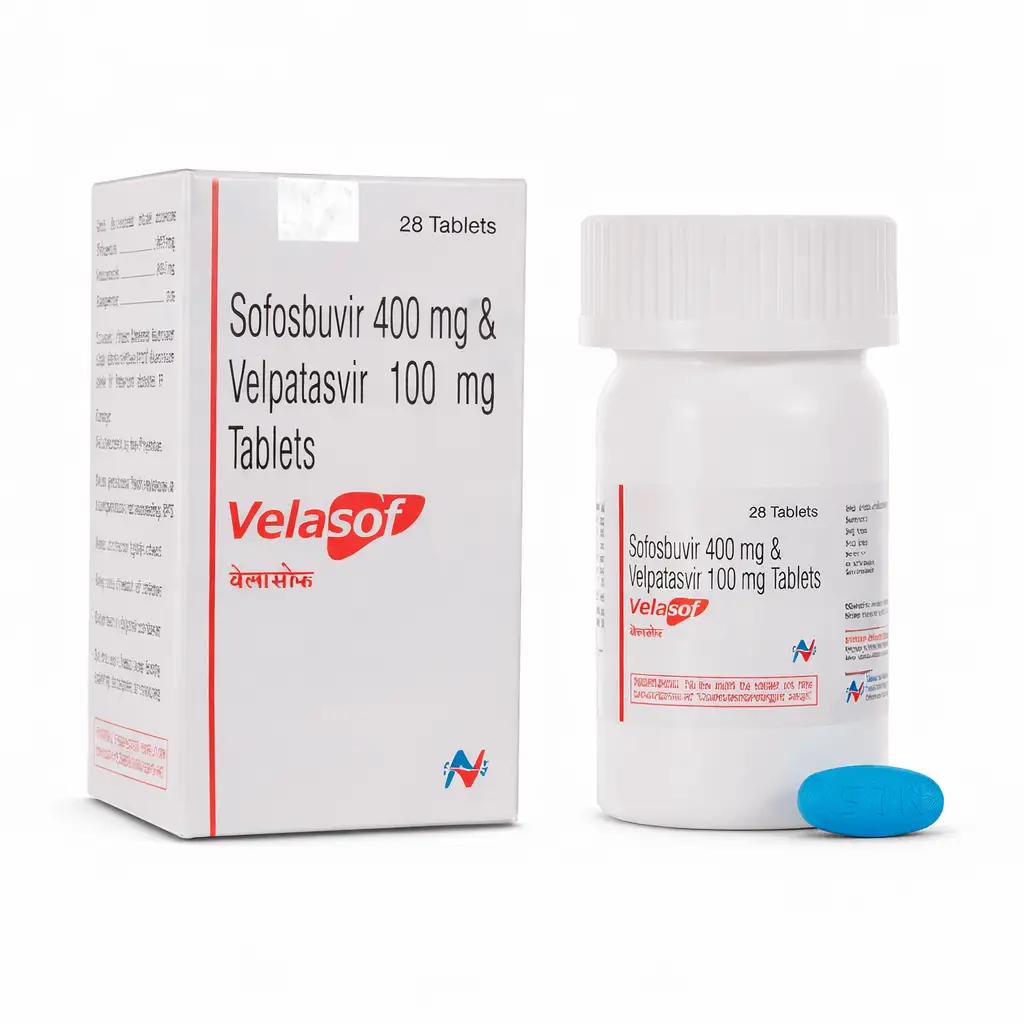
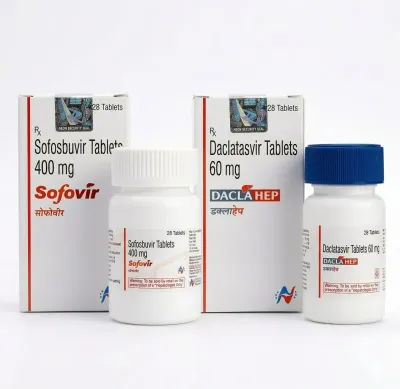
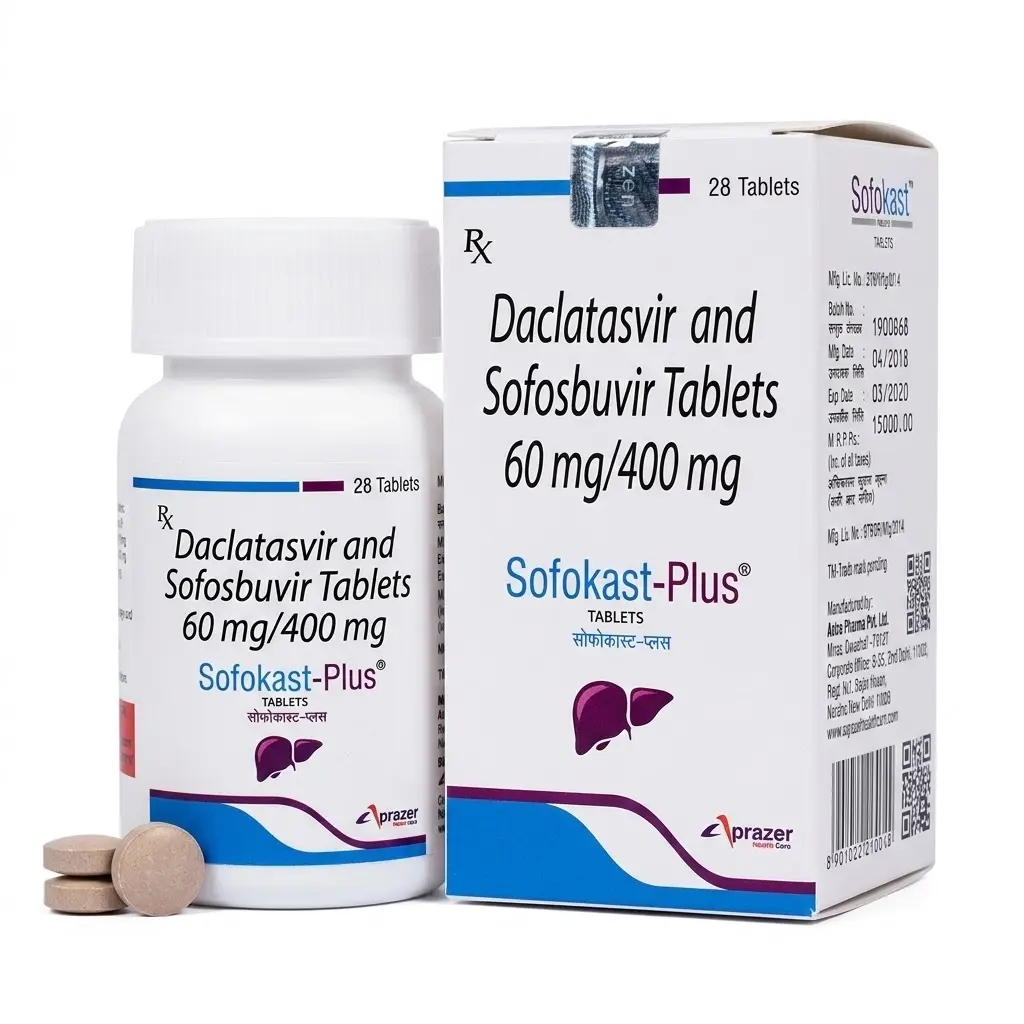
Sofosbuvir — Hepatitis C Treatment
Sofosbuvir is an innovative direct-acting antiviral (DAA) agent, which is a nucleotide analog that inhibits the NS5B RNA polymerase of the hepatitis C virus (HCV). The drug became a revolutionary solution in hepatology, enabling a transition from interferon-based regimens to entirely oral therapy with unprecedentedly high cure rates.
The mechanism of action of sofosbuvir is based on its ability to act as a false substrate for the NS5B viral polymerase. Upon entering hepatocytes, the drug undergoes intracellular metabolism to form an active triphosphate. This metabolite is incorporated into the building HCV RNA strand, causing immediate chain termination and halting viral replication. Due to its high specificity for viral enzymes, sofosbuvir exhibits minimal toxicity to human cells.
The drug is effective against all major HCV genotypes (1–6). Sofosbuvir possesses a high genetic barrier to the development of resistance, making it a foundational component of most modern combined antiviral therapy regimens.

Indications
Sofosbuvir is used for the treatment of chronic hepatitis C as part of comprehensive therapy regimens:
- Chronic Hepatitis C (Genotypes 1, 2, 3, 4, 5, 6): treatment of adult patients and pediatric patients (aged 3 years and older) in combination with other antiviral agents.
- Patients with Cirrhosis: therapy for individuals with compensated or decompensated liver cirrhosis (in combination with ribavirin).
- HIV/HCV Co-infection: treatment of patients simultaneously infected with human immunodeficiency virus and hepatitis C.
- Transplant Waitlist: prevention of HCV reinfection after liver transplantation in patients with hepatocellular carcinoma.
Dosage and administration
The sofosbuvir administration regimen is typically standardized and depends on the specific combination of drugs used.
- Standard Dose: 400 mg (one tablet) once daily.
- Administration Rules: the tablet should be taken with food and swallowed whole (it has a very bitter taste, so chewing is not recommended).
- Duration of Course: usually 12 or 24 weeks, depending on the viral genotype and the degree of liver damage (presence of cirrhosis).
- Missed Dose: if less than 18 hours have passed since the missed dose, it should be taken immediately. If more than 18 hours, wait for the next scheduled dose.
- Vomiting: if vomiting occurs within 2 hours of administration, an additional tablet should be taken.
Frequently Asked Questions
List of medicines by active substance Sofosbuvir